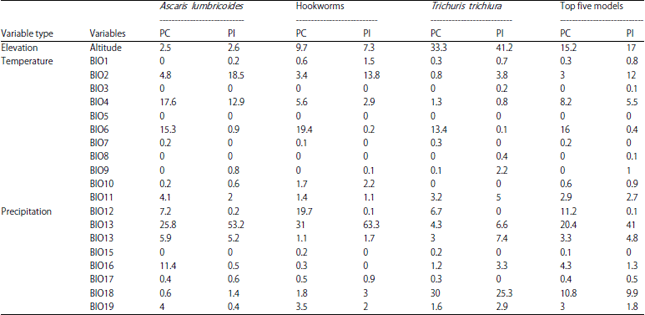
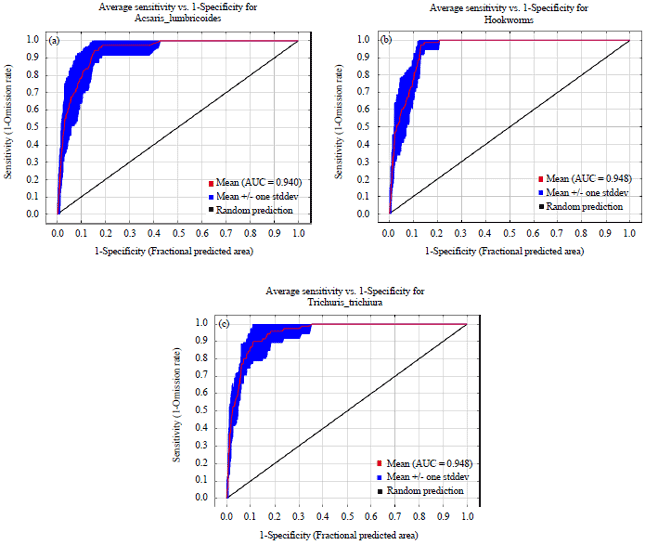
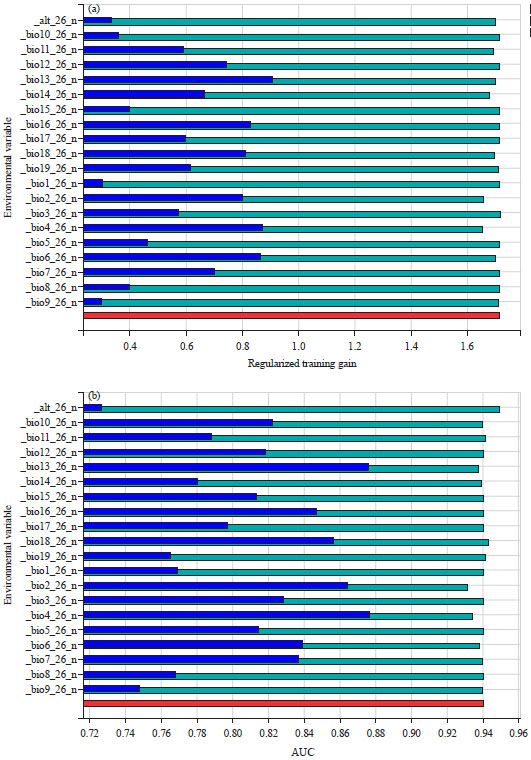
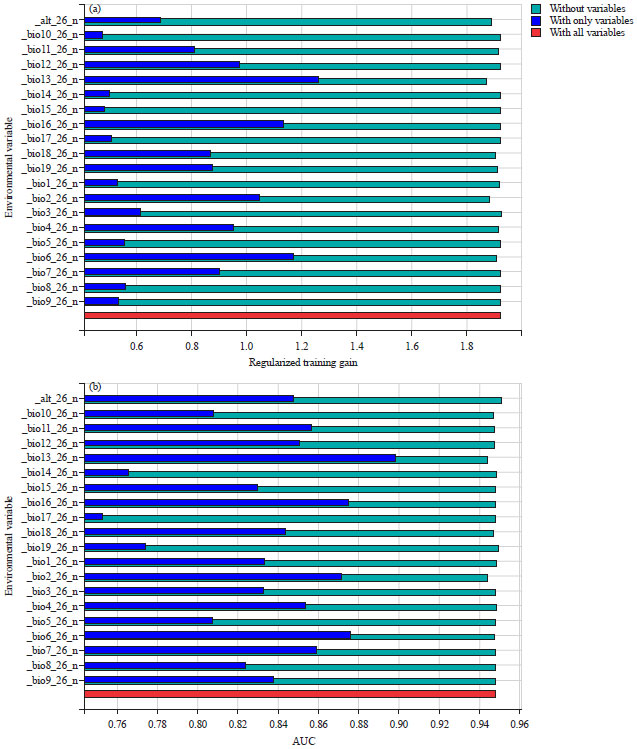
Research Article
Spatial Distribution and Modelling of Soil Transmitted Helminthes Infection in Nigeria
Department of Zoology and Environmental Biology, Kogi State University, Anyigba, Kogi State, Nigeria
LiveDNA: 234.16631
Ezekiel Kogi
Department of Zoology, Ahmadu Bello University, Zaria, Nigeria
Sodangi Abdulkarim Luka
Department of Zoology, Ahmadu Bello University, Zaria, Nigeria