Research Article
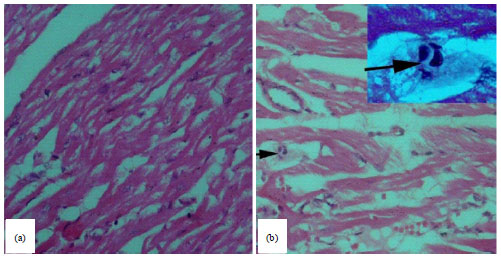
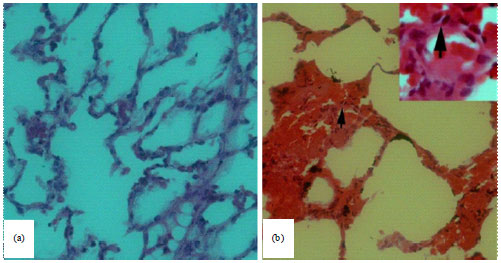
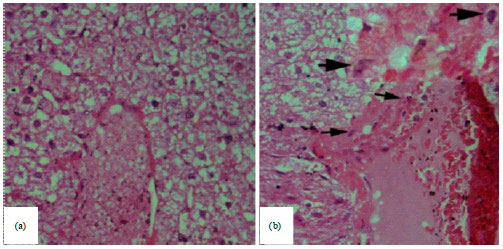
Detection of Toxoplasma gondii (Apicomplexa: Sarcocystidae) in the Brown Dog Tick Rhipicephalus sanguineus (Acari: Ixodidae) Fed on Infected Rabbits
Department of Parasitology and Animal Diseases, Veterinary Research Division, National Research Center, Giza, Egypt
Rafaat M. Shaapan
Department of Zoonotic Diseases, Veterinary Research Division, National Research Center, Giza, Egypt
Khaled A. Abdelrahman
Department of Parasitology and Animal Diseases, Veterinary Research Division, National Research Center, Giza, Egypt
Amira H. El- Namaky
Department of Parasitology and Animal Diseases, Veterinary Research Division, National Research Center, Giza, Egypt
Faten A.M. Abo- Aziza
Department of Parasitology and Animal Diseases, Veterinary Research Division, National Research Center, Giza, Egypt
Hala A. Abou Zeina
Department of Parasitology and Animal Diseases, Veterinary Research Division, National Research Center, Giza, Egypt