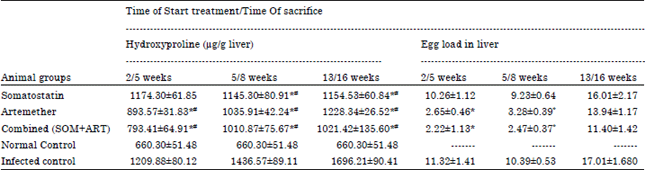
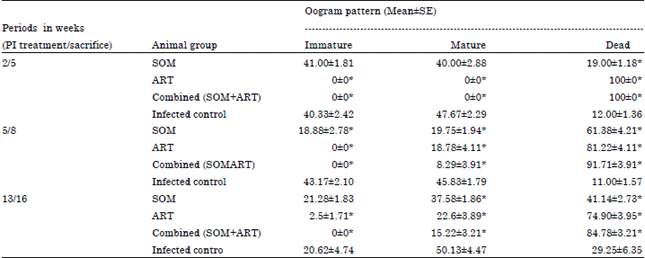
Research Article
Efficacy of Combined Therapy of Artemether and Somatostatin in Heptic Hydroxyproline in Experimental Murine Schistosomiasis Mansoni
Department of Parasitology, Faculty of Medicine for Girls, Al-Azhar University, Cairo, Egypt
Madiha Mahmoud
Department of Parasitology, Faculty of Medicine for Girls, Al-Azhar University, Cairo, Egypt