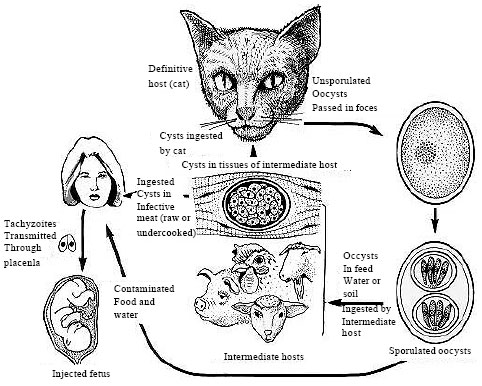
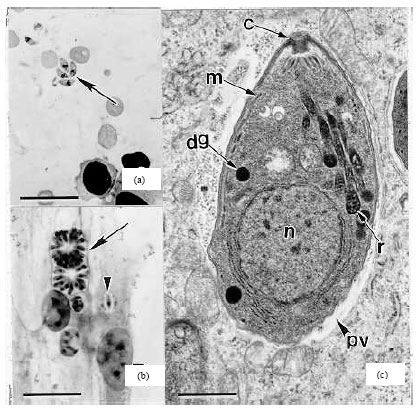
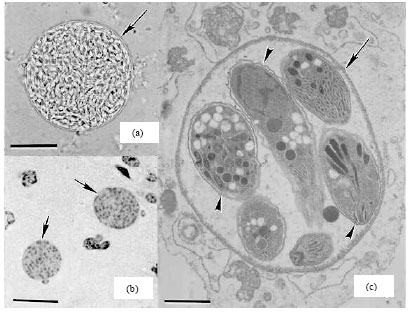
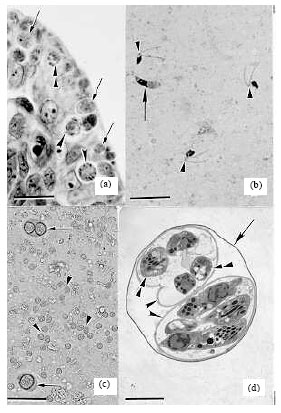
Review Article
Toxoplasma gondii Strategy for Intracellular Survival: Is it Still Enigmatic?
Department of Zoology, College of Sciences, Female Center for Scientific and Medical College, King Saud University, P.O. Box 22452, Riyadh 11495, Saudi Arabia
Ebtesam M. AL Olayan
Department of Zoology, College of Sciences, Female Center for Scientific and Medical College, King Saud University, P.O. Box 22452, Riyadh 11495, Saudi Arabia