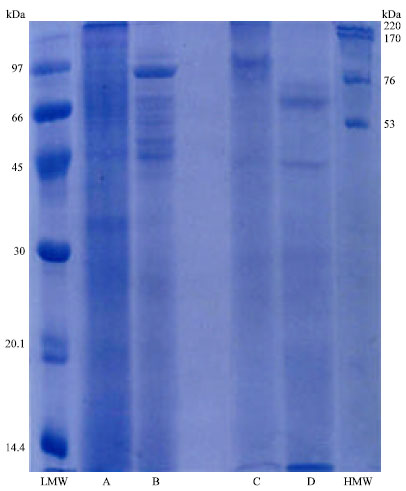
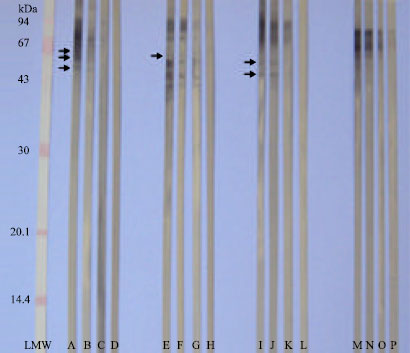
Research Article
Specificity of Snail Derived Antigens in Diagnosis of their Trematode Parasites
Department of Parasitology and Animal Diseases, National Research Center, P.O. Box 12622, Giza, Egypt
F.M. El-Moghazy
Department of Biology, College of Education for Girls- Al-Kharj, Scientific Departments, Al-Kharj University, Kingdom of Saudi Arabia
F.A.M. Khalil
Department of Parasitology and Animal Diseases, National Research Center, P.O. Box 12622, Giza, Egypt