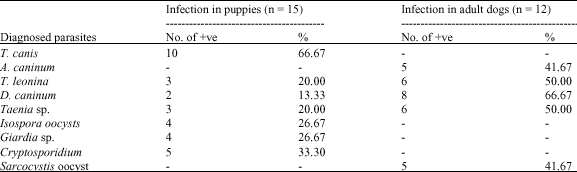
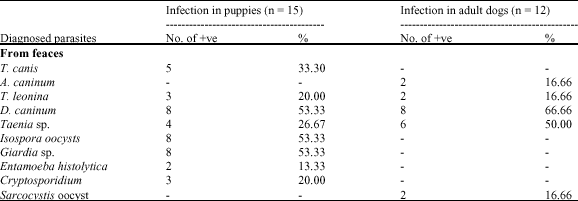
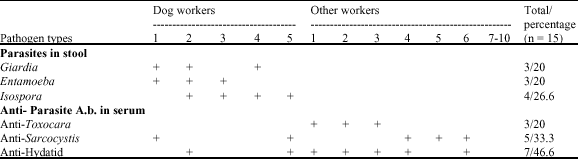
Research Article
Captive Dogs as Reservoirs of Some Zoonotic Parasites
Department of Zoonoses, Faculty of Veterinary, Medicine Cairo University, El-Giza, Egypt
Hossein S. Lotfy
Department of Parasitology, Animal Health Research Institute, Beni-Suef Branch, Beni Suef, Egypt