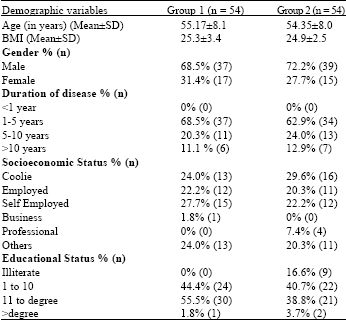
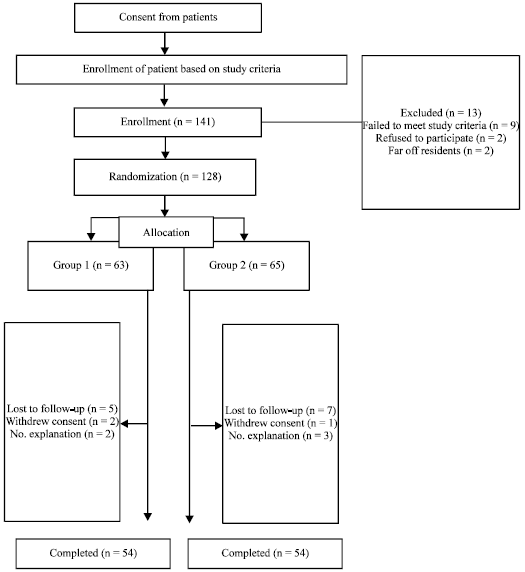
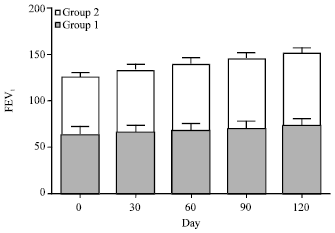
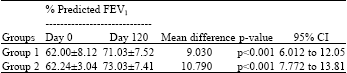Research Article
Effect of Two Controller Medications with Inhaled Corticosteroid in Mild to Moderate Persistent Asthma Patients
Department of Pharmacy Practice, SRM College of Pharmacy, SRM University, Kattankulathur-603 203, Kanchipuram (DT), Tamil Nadu, India
A.D. Nageswari
Department of Pulmonary Medicine, SRM Medical College Hospital and Research Centre, SRM University, Kattankulathur-603 203, Kanchipuram (DT), Tamil Nadu, India
K. Ilango
Interdisciplinary School of Indian System of Medicine (ISISM), SRM University, Kattankulathur-603 203, Kanchipuram (DT), Tamil Nadu, India