Research Article
Prevalence and Antimicrobial Resistance of Salmonella Isolated from Raw Milk Samples Collected from Kersa District, Jimma Zone, Southwest Ethiopia
Department of Biology Jimma University P.O Box 378, Jimma, Ethiopia
Anbessa Dabassa
Metu university Biology department, Metu, Ethiopia










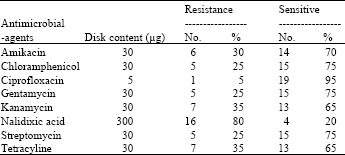
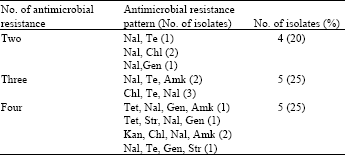
shubisa abera leliso Reply
interested to read