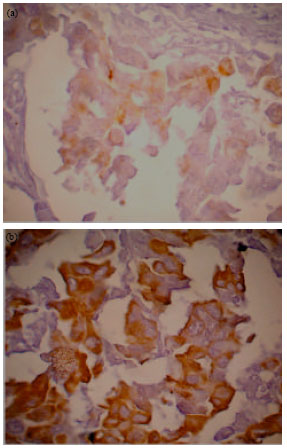
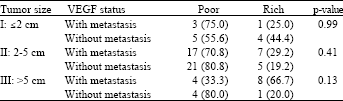
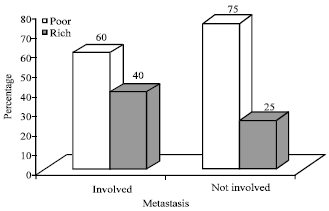
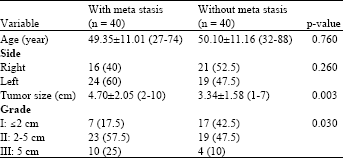Research Article
Association of VEGF with Regional Lymph Node Metastasis in Breast IDC
Department of Pathology, Tabriz University of Medical Sciences, Imam Reza Hospital, Tabriz, Iran
Amir Vahedi
Department of Pathology, Tabriz University of Medical Sciences, Imam Reza Hospital, Tabriz, Iran
Ebrahim Kord Mostafapour
Tehran University of Medical Sciences, Tehran, Iran