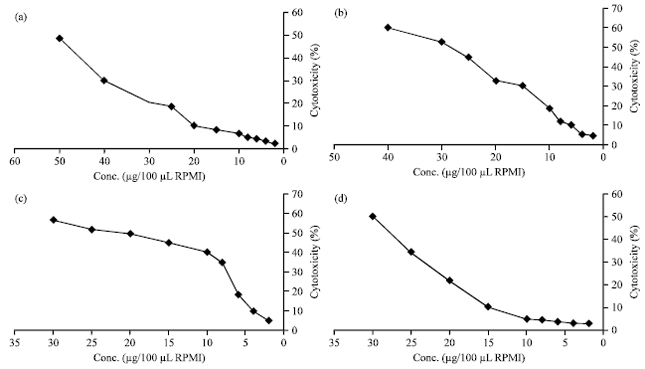
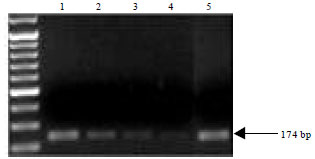
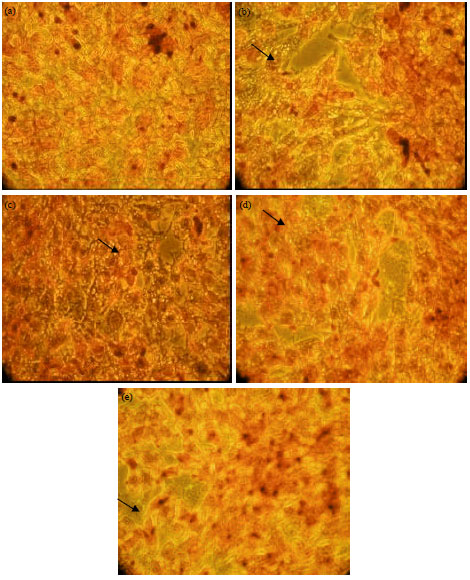
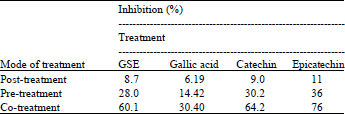
Research Article
The Potentiality of Grape Seed Extract as a Novel Anti-hepatitis C virus Agent
Department of Protein Researches, Genetic Engineering and Biotech Institute, City for Scientific Research, New Borg Elarab, 21934 Alexandria, Egypt
Nehal M. El-Deeb
Department of Medical Biotechnology, Genetic Engineering and Biotech Institute, City for Scientific Research, New Borg Elarab, 21934 Alexandria, Egypt
Hala I. EL-Adawi
Department of Medical Biotechnology, Genetic Engineering and Biotech Institute, City for Scientific Research, New Borg Elarab, 21934 Alexandria, Egypt