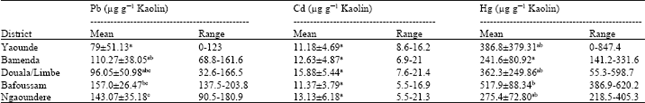
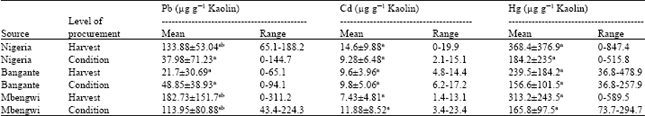
Research Article
Intake of Lead, Cadmium and Mercury in Kaolin-eating: A Quality Assessment
Food and Nutrition Research Centre, P.O. Box 6163, Yaounde, Cameroon
C.M. F. Mbofung
National School of Agro-Industrial Sciences, P.O. Box 686, Ngaoundere, Cameroon
D. N. Lantum
FMBS, P.O. Box 1364, Yaounde, Cameroon