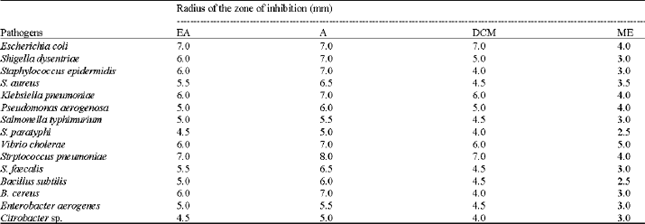
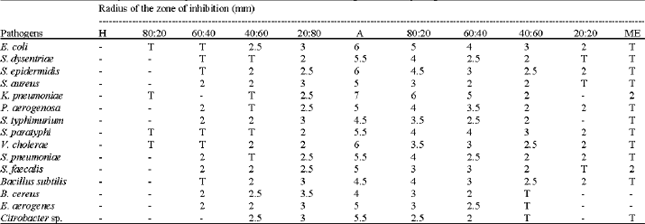
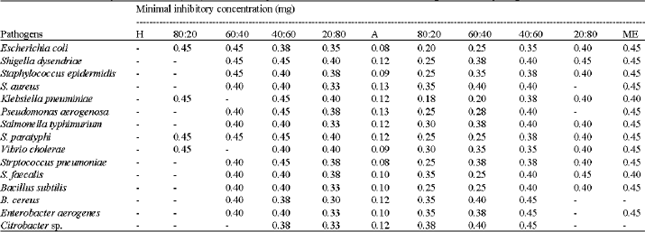
Research Article
Bioactive Potential of Coral Associated Gastropod, Trochus tentorium of Gulf of Mannar, Southeastern India
Department of Industrial Biotechnology, Bharath University, Chennai, Tamil Nadu, India
C. Chellaram
Department of Biomedical Engineering, Veltech Multitech Dr. R.R. Dr. S.R. Engineering College, Chennai-600 062, Tamil Nadu, India
S. Jonesh
Department of Biomedical Engineering, Veltech Multitech Dr. R.R. Dr. S.R. Engineering College, Chennai-600 062, Tamil Nadu, India
L. Jayanthi
Department of Industrial Biotechnology, Bharath University, Chennai, Tamil Nadu, India
J.K.P. Edward
Suganthi Devadason Marine Research Institute, Tuticorin-628 001, Tamil Nadu, India