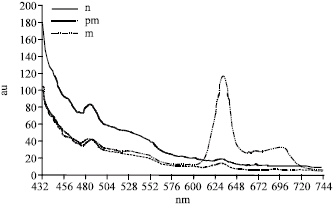
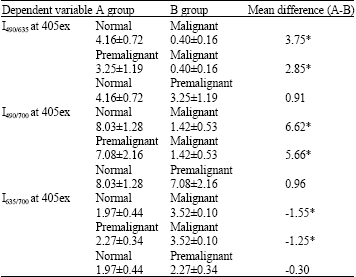
Research Article
Autofluorescence Spectroscopy of Oral Squamous Cell Carcinoma
Department of Oral and Maxillofacial Pathology, Rajah Muthiah Dental College and Hospital, Annamalai University, Annamalainagar-608 002, Tamil Nadu, India
S. Jayalakshmi
CAS in Marine Biology, Annamalai University, Parangipettai-608 502, Tamil Nadu, India
C.R. Ramachandran
Department of Oral and Maxillofacial Pathology, Rajah Muthiah Dental College and Hospital, Annamalai University, Annamalainagar-608 002, Tamil Nadu, India