Research Article
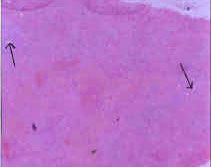
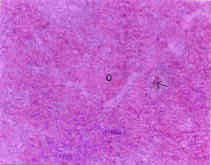
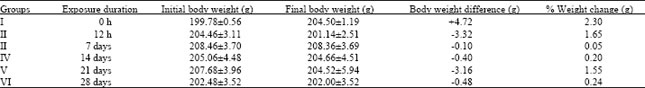
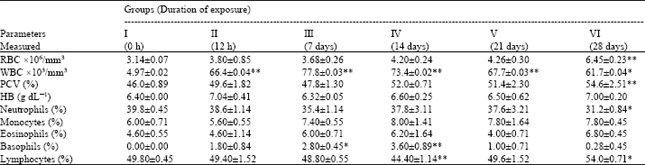
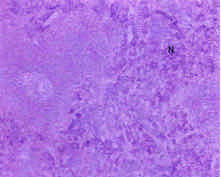
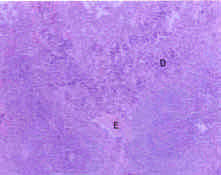
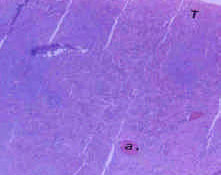
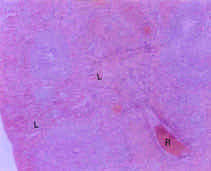
Toxicological Effects of Inhaled Mosquito Coil Smoke on the Rat Spleen: A Haematological and Histological Study
Department of Human Anatomy: College of Medical Sciences, P.M.B. 1069, University of Maiduguri, Nigeria
M.M. Shehu
Department of Human Anatomy: College of Medical Sciences, P.M.B. 1069, University of Maiduguri, Nigeria
A.B. Adelaiye
Department of Human Physiology, Faculw of Medical Sciences, Ahmadu Bello University, Zaria, Nigeria