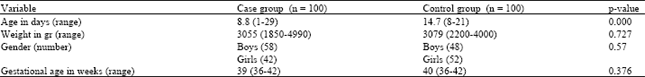
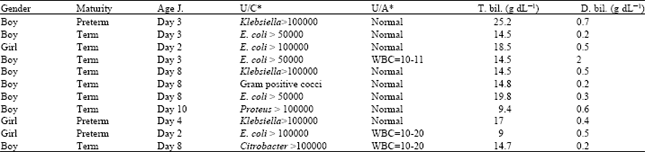
Research Article
Investigation of Urinary Tract Infection in Neonates with Hyperbilirubinemia
Department of Pediatric, NICU, Shahid Sacloughi University of Medical Sciences, Yazd, Iran
Mohammad Hasan Sheikhha
Department of Genetic, Clinical and Research Center for Infertility, Shahid Sadoughi University of Medical Sciences, YazcL Iran