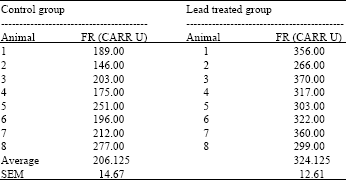
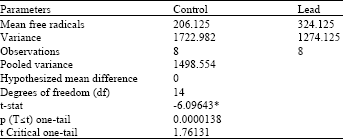
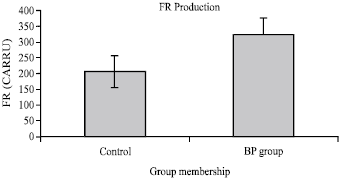
Research Article
Effects of Lead Toxicity on Free Radical Production in Rats
Department of Zoology, College of Science, King Saud University, P.O. Box 2455, Riyadh, 1 1451, Saudi Arabia
A. S.A. Haffor
Department of Zoology, College of Science, King Saud University, P.O. Box 2455, Riyadh, 1 1451, Saudi Arabia