Research Article
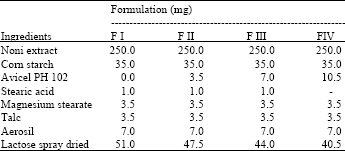
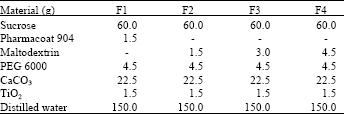
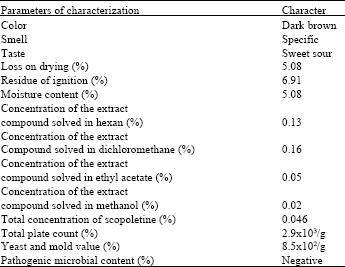
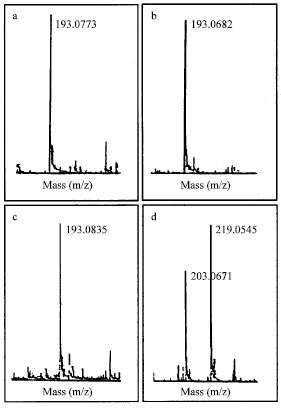
Study of Coating Tablet Extract Noni Fruit(Morinda citrifolia, L.) with Maltodextrin as a Subcoating Material
Department of Pharmacy, Faculty of Mathematics and Science, University of Indonesia, Kampus UI Depok, 16424, Indonesia
Arsyadi .
Naval Pharmacy Institute, Jl. Bendungan Jati Luhur Xo. 1 Jakarta Pusat, 10210, Indonesia
L. Broto S. Kardono
Indonesian Science Institute-LIPI, Puspitek, Servong, 15314, Indonesia