ABSTRACT
Immunohistochemical staining was undertaken for E-cadherin (EC), estrogen and progesterone receptors; on tissue sections of normal breast, proliferative breast lesions and malignant breast lesions. The EC positive expression manifested itself as sharp membranous staining and was assessed semi-quantitative into four categories: 0; 1+ were considered negative immunoreactivity, while 2+ and 3+ were scored as positive immunoreactivity. Although 95% of non malignant proliferative breast lesions showed positive EC immunoreactivity and reduced or lost EC expression in all Pre-invasive cases. None of invasive lobular cases express EC, in contrast to 30% of invasive duct carcinoma cases, showed reduced or lost EC expression in high histological grades. Correlation between the EC staining intensity and IDC, ILC and TC groups, statistical analysis was highly significant only for TC (p< 0.001). There was a significant relationship between E-cadherin expression and different breast neoplastic histological types. The molecular signature of breast lobular carcinomas is the loss of E-cadherin protein expression as evidenced by immunohistochemistry, whereas ductal carcinomas are typically E-cadherin positive. Present study suggests that E-cadherin may be involved in the pathogenesis of this form of breast cancer.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jms.2007.740.747
URL: https://scialert.net/abstract/?doi=jms.2007.740.747
INTRODUCTION
The three most commonly diagnosed cancers among women in 2006 will be cancers of the breast, lung and bronchus and colon and rectum, accounting for about 54% of estimated cancer cases in women (Jamal et al., 2006). Breast cancer alone is expected to account for 31% of all new cancer cases among women. Human breast carcinomas most frequently evolve from the epithelial lining of the terminal mammary ducts that may progressively become invasive and ultimately metastatic (Anonymous, 2002; O`Shaughnessy, 2006). The transformation of normal mammary epithelial cells into a carcinoma and the subsequent progression to invasion and metastasis involve the accumulation of numerous genetic hits, including the activation or amplification of dominant oncogenes and the deletion or inactivating mutation of key tumor suppressor genes (Goldstein, 2002; Graff et al., 2000). Absent cell-to-cell adhesion seems to be a necessary property of carcinoma cells to facilitate permeation through tissue planes and produce characteristic lobular carcinoma-type (Handschuh et al., 1999; Rashid et al., 2001). An observation of interest was that the expression of E-cadherin requires further evaluation for confirmation of a common regulatory pathway that could be activated in the early onset of nodal metastasis (Pinder et al., 1998; Vos et al., 1997). E-cadherin expression is diverse and differences in patient characteristics may produce variability in expression. E-cadherin is a transmembrane glycoprotein that mediates epithelial cell-to-cell adhesion (Acs et al., 2001). E-cadherin (EC) is a calcium-regulated adhesion molecule expressed in most normal epithelial tissues Whereas some studies have indicated that down regulation of e-cadherin, associated with loss of cellular adhesiveness, was correlative with poor prognosis and metastasis, other studies have failed to confirm this (Charpin et al., 1998; Guriec et al., 1996; Hunt et al., 1997). The present study examine the relationship between E-cadherin and other prognostic markers in breast cancer. The ER and the E-cad gene have been implicated frequently in the initiation and/or progression of human breast cancer (Bracke et al., 1996; Kanai et al., 1994; Nass et al., 2000). Loss of expression of either gene has been associated with poorly differentiated tumors and poorer prognosis (Larue et al., 1994; Berx et al., 1995a, b; Heimann et al., 2000; Siitonen et al., 1996). Furthermore, several studies have reported an association between E-cad and ER expression in breast tumors (De Leeuw et al., 1997a, b) Because loss of E-cadherin expression results in disruption of cellular clusters, it has been postulated that E-cadherin functions as a tumor suppressor protein (Gumbiner, 2000; Rubin et al., 2001). The role of E-cadherin in highly aggressive form of breast cancer, is largely unknown (Pierceal et al., 1995; Umbas et al., 1994; Umbas et al., 1997). Adhesion molecules, particularly cadherins play a pivotal role in cancer invasion and metastasis (Gumbiner, 2000; Rubin et al., 2001). Because the therapeutic management of tumors with and without nodal metastasis differs considerably (Heimann et al., 2000). Tumor cells strongly express E-cadherin, thereby providing an important exception to the positive association between E-cadherin loss and poor prognosis in breast cancer (Anonymous, 2002; O`Shaughnessy, 2006). The objective of this study was to assess whether E-cadherin expression contributes to the development and progression of different breast carcinomas and to investigate any differences in the expression of E-cadherin immunohistochemically in different cases of breast carcinomas. Histopathological study was, on formalin-fixed, paraffin-embedded tissue sections and immunostained for E-cadherin, estrogen and progesterone receptors (ER and PR, respectively). We have immunohistochemically investigated E-cadherin (E-CD) expression in a series of relationships between membrane E-cadherin reactivity of invasive carcinoma, a dyshesive growth pattern and lobular carcinoma-type Infiltrating Ductal Carcinomas (IDC) in an attempt to assess the biological and prognostic relevance of E-CD in patients harboring breast carcinomas. We evaluated the EC expression as an aid to subclassification of invasive breast carcinoma. And correlated with various clinical and pathological prognostic factors: the histologic type, grade status, tumor size, hormone receptor status (ER and PR) Immunoreactivity for estrogen receptor, progesterone receptor (in >10% of lesional cells).
MATERIALS AND METHODS
Retrieved 50 different breast lesions and breast carcinomas cases from the surgical pathology files of Kasr EL Aini Hospital in Cairo University in the period 2001 to 2002. The H and E-stained slides were reviewed to grade and subclassify the tumors based on established criteria without knowledge of immunohistochemical results. Primary tumour size was reported based on TNM system. Five micron thick sections were obtained from 10% formalin fixed tissue samples paraffin-embedded tissue samples for routine heamatoxylin and eosin stain. Histological grading after final histologic review using the Elston/Nottingham modification of Bloom-Richardson system according to Bane et al. (2005).
Cases were selected for study, including non malignant breast lesions 3 cases of duct papilloma, 5 cases of fibroadenoma, 3 cases of lobular hyperplasia. and malignant breast lesions including 5 cases of Duct Carcinoma in situ (DCIS), 4 cases of Lobular Carcinoma Insitu (LCIS), 18 cases of Invasive Duct Carcinoma (IDC), 8 cases of Invasive Lobular Carcinoma (conventional ILC), 4 cases of Tubular Carcinoma (TC). Data on tumor size, axillary lymph node status (axillary lymph nodes from radical mastectomy specimens), stage of disease, were abstracted from the pathology reports.
Immunohistochemical analysis was performed using a monoclonal antibody (clone 4A 27, Zymed, South San Francisco, CA) to EC on formalin-fixed, paraffin-embedded specimens were cut and mounted onto positive charged glass slides. Samples were deparaffinized and rehydrated before incubation for 5 min with 5% hydrogen peroxide to inhibit endogenous peroxidase. The tissue sections were treated using heat induced antigen retrieval technique. After treatment with blocking serum (to reduce the nonspecific binding of conjugated second antibody), the samples were incubated for 60 min with monoclonal antibody anti-E-Cadherin second generation prediluted antibody. Then rinsed well with Phosphate Buffer Saline (PBS). Two drops of biotinylated secondary product was put on the sections and incubated for 10 min then rinsed with PBS. Two drops of enhanced horseradish peroxidase conjugated streptavidine were added to each section, incubated for 10 min then rinsed well with PBS. The presence of peroxidase was revealed by addition of diaminobenzidine (DAB) chromogen that creates an intense brown deposit around the antigen/antibody/ enzyme complex. Finally sections were counter stained with Mayer`s hematoxylin and mounted. Immunostains were evaluated independently and any differences in interpretation were resolved by simultaneous viewing. Evaluation of immunostaining and scoring was performed by light microscopy. Positive expression of the E-cadherins manifested itself as sharp membranous staining and with cytoplasmic staining in a few lesions and was statistically significant (Bukholm et al., 2000). Only the membrane staining intensity and pattern was evaluated on a scale of 0 to 3, scores of 0 and 1+ were considered negative immunoreactivity of 2+ and 3+ was scored as positive. Cytoplasmic staining was rare, considered nonspecific and not included in assessment. The presence of EC staining in epithelial cells of normal ducts and acini served as an internal positive control in every case (Bukholm et al., 2000). Negative controls in which the primary antibody was replaced with tris-buffered saline, were included in every run. All sections were first screened to disclose the areas with well preserved tissue architecture and cell morphology for scoring of immunoreactivity. Necrotic areas were discarded in the analysis count mitotic figures at periphery of tumor in most mitotically active area; count 10 high power fields in the same area, but necessarily contiguous; select fields with as much tumor as possible; avoid poorly preserved areas.
The immunostain scores were correlated with the histologic type, grade status, tumor size, hormone receptor status (ER and PR) Immunoreactivity for estrogen receptor, progesterone receptor (in >10% of lesional cells). The association between EC and tumor type was assessed by using the chi square test. Associations with ER and PR were assessed. A 2-sided p-value less than 0.05 was considered statistically significant.
RESULTS
Histological study and tumor subtypes, tumor grades along with EC immunoreactivity are summarized in Table 1.
Histologic types and E-cadherin: Correlation between E-cadherin intensity and histological grades of IDC cases studied in Table 2.
IDC and ductal special types: Positive EC expression was seen in all but 6(33.3%) cases of IDC (Fig. 1, 2 and 4; Table 1 and 2) and all 4 ductal special types (Tubular Carcinoma TC) (100%). EC expression was present in 100% of tumor cells in all positive cases and the staining was 2+ in 5 cases and 3+ in only 3 cases (Fig. 1, 3 and 4; Table 3 and 4).
| Table 1: | Histologic tumor subtypes and tumor grade along with EC immunoreactivity |
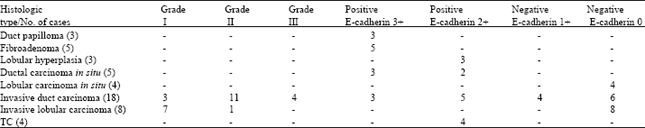 | |
| All invasive carcinomas were graded using The Elston/Nottingham modification of Bloom-Richardson grading system | |
| Table 2: | Correlation between E-cadherin intensity and histological grades of IDC cases |
 | |
| Table 3: | Analysis of prognostic tumor parameters with loss of E-cadherin expression in all carcinomas (data are given as percentage) |
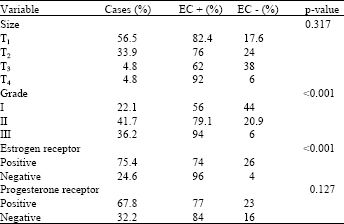 | |
| p-value less than 0.05 was considered statistically significant | |
The all 4 cases of tubular carcinoma showed 3+ EC positive immunoreactivity (Fig. 4). Associated Ductal Carcinoma in situ (DCIS) was positive with 3+ EC immunoreactivity (Fig. 2).
Invasive lobular carcinoma: Of 8 ILCs with the classic histologic pattern, (100%) showed complete loss of EC; 2 histologically typical ILC (25%) showed faint incomplete membrane staining in less than 10% of tumor cells. Seven of these EC-negative cases were well-differentiated (nuclear grade I). Six cases IL showed loss of EC membrane staining in invasive and corresponding in situ components (Fig. 3 and 4; Table 1 and 2).
Statistical analysis
E-cadherin expression and tumor subtype: All nonmalignant proliferative breast lesions included in this study showed strong (3+) positive EC immunoreactivity and all cases of Duct Papilloma and fibroadenoma.
All cases of Lobular Hyperplasia showed weak (2+) positive EC immunoreactivity (Fig. 2 and Table 1). Comparison of EC staining in IDC, ILC and Tubular carcinoma (Table 1, Fig. 1, 3 and 4) TC revealed a highly statistically significant difference between the groups (p<001). Overall, negative staining of EC in ILC was specific for the diagnosis of ILC (Fig. 3).
E-cadherin expression versus tumor parameters: All Invasive Carcinomas EC expression and tumor characteristics did not reveal statistically significant associations with loss of EC expression However, loss of EC was associated significantly with tumor grade (Table 3; Fig. 1 and 4).
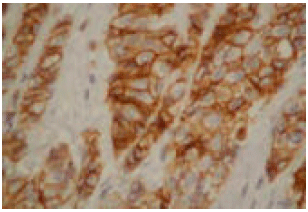 | |
| Fig. 1: | Invasive Ductal Carcinoma (IDC) grade I, with strong 3 + positive immunoreactivity to E-cadherin (x200) |
All carcinomas excluding ILC grade, size and hormonal status. Complete loss of EC was seen in 6 cases of IDC and special types to be of prognostic or predictive value (Table 3). Although only (44%) of IDC expressed E-CD in >5% of tumor cells (EC-positive carcinomas), of higher histological grade (p = 0.001). In addition, E CD-positive tumors were negative for estrogen (p = 0.001) and progesterone receptors (p = 0.001) and showed reduced E-cadherin expression (p = 0.276) more frequently than E-CD-negative tumors.
DISCUSSION
In this study normal ductal and acinar epithelium, expressed EC strongly as was also demonstrated by Bukholm et al. (1998 and 2000). All nonmalignant proliferative breast lesions included in this study showed strong (3+) positive EC immunoreactivity as well as all cases of Duct Papilloma and fibroadenoma. All cases of lobular hyperplasia showed weak (2+) positive EC immunoreactivity (Table 1, Fig. 1 and 2). According to Bukholm et al. (2000) demonstrated a similar EC expression in the normal ducts and lobular epithelium and also in ductal hyperplasia, but reported marked loss of EC in Atypical lobular Hyperplasia.
In the present study all cases of LCIS showed total loss of EC expression and DCIS showed either weak or strong EC expression (40 and 60%, respectively) (Table 1, Fig. 2 and 3). Similar results have been recorded by Bukholm et al. (2000), where total loss of EC expression was found in all cases of LCIS cases, whereas, DCIS cases almost exhibited EC expression. Loss of EC expression was noticed by Gumbiner (2000).
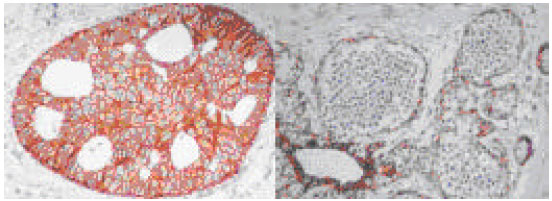 | |
| Fig. 2: | Duct carcinoma in situ (DCIS) with strong 3+ positive immunoreactivity to E-cadherin (to the left), versus the negative staining in case of lobular carcinoma in situ (LCIS) to the (right); with the intact peripheral myoepithelium preserved and served as positive internal control (x200) |
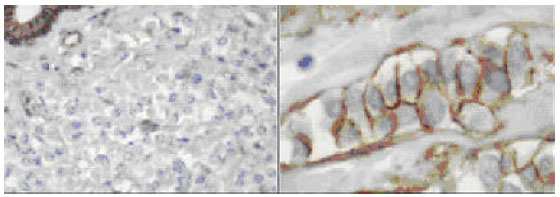 | |
| Fig. 3: | E-cadherin immunostains can aid in the distinction of lobular carcinoma, EC doesn’t show significant staining of the membrane (to the left) in contrast to ductal carcinoma grade I (to the right). Invasive duct carcinoma grade I with strong 3+ positive immunoreactivity to E-cadherin. A benign duct serves as a positive internal control (x400) |
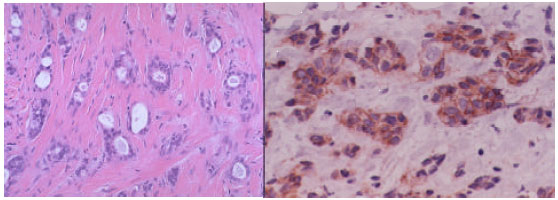 | |
| Fig. 4: | Tubular carcinoma showing irregular tubular glands arranged haphazardously in a fibrotic stroma without a lobular pattern. Coexisting cribriform ductal carcinoma in situ is seen in the right, H and E (x100) to the left. Tubular carcinoma with strong 3+ positive E-cadherin immunoreactivity (x200) to the right |
Furthermore, Acs et al. (2001) found no expression of EC not only in ILC and LCIS adjacent to ILC but also in LCIS without an invasive component indicating a role for EC mutation even before tumour invasion. Whereas EC expression was present or reduced in DCIS, they concluded that EC is a classical tumour-suppressor gene and a very early target in lobular carcinogenesis (Pinder et al., 1998; Wahed et al., 2002).
Umbas et al. (1994 and 1997) used the term Lobular carcinoma in situ for a special type of noninvasive carcinoma of breast associated with a monotonous intralobular proliferation of cells.
The concurrent invasive carcinoma with absence of tubule formation and single-file growth pattern was established as ILC. (Gumbiner, 2000; Sarrio et al., 2003; Wahed et al., 2002). The distinctive histologic features of this special type of breast carcinoma described by Jones and Laird (1999) and Graff et al. (2000) paved the way for identification of this tumor by pathologists when the classic features are present. Identification of variants of ILC has added new dilemmas to the existing problem of distinguishing IDC of no special type with cord-like or trabecular patterns from ILC and its variants (Singletary et al., 2002; Woodward et al., 2003).
Selective EC loss, now well recognized, validates ILC as a distinct entity and explains its histologic appearance, (Jones and Laird, 1999; Graff et al., 2000) and distinctive growth patterns in metastases (Singletary et al., 2002; Woodward et al., 2003).
Although EC is emerging as an excellent marker to type breast carcinomas (Jones and Laird, 1999; Graff et al., 2000) the conflicting reports of EC loss as predictor of increased invasiveness, metastatic potential and poor survival (Jones and Laird, 1999; Graff et al., 2000) raise questions about its reliability for typing. Loss of EC alone cannot be a predictor of metastatic potential and negative outcome as EC is lost even in the preinvasive stages of LCIS and atypical lobular hyperplasia (Jones and Laird, 1999; Graff et al., 2000; Singletary et al., 2002; Woodward et al., 2003).
Furthermore, ILC is a slow-growing tumor that has been shown to have better survival than ductal no special type (Singletary et al., 2002; Woodward et al., 2003).
As demonstrated in this study and in previous studies by De Leeuw et al. (1997); Acs et al. (2001) and Wahed et al. (2002) that EC can help in the diagnosis of ILC. In this study, complete EC loss is reported in 100% of ILCs, as reported also by Singletary et al. (2002) and Woodward et al. (2003) showing good membrane positivity in all cases of IDC, including the special type Tubular Carcinoma (Fig. 1, 2 and 4, Table 1, 2 and 3). Most of the ductal invasive and in situ carcinomas cases showed strong EC-positive (3+) immunoreactivity (Fig. 1, 2 and 4). The exception was in the 4 cases of TC that showed 2+ staining in the tubules only (Fig. 4) and a very few high-grade cellular IDCs with apparent reduced expression of EC (Fig. 1, 2 and 3).
Berx et al. (1995) and Acs et al. (2001) observed variation in EC intensity in IDC, diagnostic difficulty occurs in some cases because IDC may show a dispersed growth pattern, including infiltration around benign ducts in a targetoid manner similar to ILC (Sarrio et al., 2003; Woodward et al., 2003).
Most studies have observed retained EC expression in almost all IDCs but reduced expression mainly associated with poor differentiation and high tumor grade (Sarrio et al., 2003; Woodward et al., 2003). Various studies have observed a correlation between reduced EC expression and lymph node status (Charpin et al., 1998; Goldstein, 2002) and ER and PR status (Kleer et al., 2001; Nass et al., 2000; Rashid et al., 2001; Rubin et al., 2001; Seidman et al., 2001) Others have found no relationship to nodal or receptor status.
Present findings were similar to Acs et al. (2001) and Goldstein et al. (2002), proved no correlation between EC expression with tumor size, grade, tubule formation, nuclear pleomorphism, mitotic activity, ER and PR status in invasive carcinomas.
Absent cell-to-cell adhesion seems to be a necessary property of carcinoma cells to facilitate permeation through tissue planes and produce characteristic lobular carcinoma-type Bukholm et al. (2000). An observation of interest was that the expression of E-cadherin requires further evaluation for confirmation of a common regulatory pathway that could be activated in the early onset of nodal metastasis. Loss of EC is a sensitive and relatively specific marker to confirm a diagnosis of ILC and its variants. EC positivity clearly favors ductal differentiation in ambiguous cases. Partial loss of EC in a minority of poorly differentiated IDCs is not of diagnostic significance.
CONCLUSION
All nonmalignant proliferative breast lesions included in this study showed strong (3+) positive EC immunoreactivity. There was reduced or lost (EC) expression in all Pre-invasive breast carcinomas cases.
Invasive Lobular Carcinoma (ILC) and Invasive Ductal Carcinoma (IDC) cases showed striking difference in their (EC) expression. Moderate to strong membrane expression found in all invasive (100%) and in situ Ductal Carcinomas.
Loss of EC alone cannot be a predictor of metastatic potential and negative outcome, as EC is lost even in the preinvasive stages of LCIS and atypical lobular hyperplasia.
All invasive (100%) and in situ lobular carcinomas showed complete loss of E-cadherin expression. Lobular and not ductal in situ neoplasia displays loss of E-cadherin expression has greatly facilitated the categorization of a large proportion of morphologically ambiguous intraepithelial neoplasias into ductal or lobular types.
These results suggest that although E-cadherin expression may be involved in the progression of IDCs, its value as an independent prognostic factor remains to be established. The level of decreased E-cadherin expression at which a dyshesive growth pattern emerges in primary breast carcinomas may be less than the level associated with lobular carcinoma-type.
Tubular Carcinoma revealed a highly statistically significant difference between the groups (p<001). Overall, negative staining of EC in ILC was specific for the diagnosis of ILC.
Loss of EC is a sensitive and relatively specific marker to confirm a diagnosis of ILC and its variants. EC positivity clearly favors ductal differentiation in ambiguous cases. Partial loss of EC in a minority of poorly differentiated IDCs is not of diagnostic significance. Present study suggests that E-cadherin may be involved in the pathogenesis of this form of breast cancer We considered the inherent loss of EC in all lobular breast carcinoma and the expression of other prognostic tumor variables that previous studies have not considered. There was a significant relationship between E-cadherin expression and different breast lesions and carcinomas histologic types.
The molecular signature of mammary lobular carcinomas is the loss of E-cadherin protein expression as evidenced by immunohistochemistry, whereas ductal carcinomas are typically E-cadherin positive.
REFERENCES
- Acs, G., T.J. Lawton, T.R. Rebbeck, V.A. Livolsi and P.J. Zhang, 2001. Differential expression of E-cadherin in lobular and ductal neoplasms of the breast and its biologic and diagnostic implications. Am. J. Clin. Pathol., 115: 85-98.
PubMed - Berx, G., A.M. Cleton-Jansen, F. Nollet, W.J. de Leeuw, M. van de Vijver, C. Cornelisse and F. van Roy, 1995. E-cadherin is a tumour/invasion suppressor gene mutated in human lobular breast cancers. EMBO J., 14: 6107-6115.
PubMed - Bracke, M.E., F.M. Von Roy and M.M. Mareel, 1996. The E-cadherin/catenin complex in invasion and metastasis. Curr. Top. Microbiol. Immunol., 213: 123-161.
PubMed - Bukholm, I.K., J.M. Nesland and A.L. Borresen-Dale, 2000. Re-expression of E-cadherin, alpha-catenin and beta-catenin, but not of gamma-catenin, in metastatic tissue from breast cancer patients. J. Pathol., 190: 15-19.
Direct Link - Charpin, C., S. Garcia, P. Bonnier, F. Martini and L. Andrac et al., 1998. Reduced E-cadherin immunohistochemical expression in node-negative breast carcinomas correlates with 10 year survival. Am. J. Clin. Pathol., 109: 431-438.
PubMed - De Leeuw, W.J., G. Berx, C.B. Vos, J.L. Peterse and M.J. van de Vijver et al., 1997. Simultaneous loss of E-cadherin and catenins in invasive lobular breast cancer and lobular carcinomas in situ. J. Pathol., 183: 404-411.
PubMed - Goldstein, N.S., 2002. Does the level of E-cadherin expression correlate with the primary breast carcinomas infiltration pattern and type of systemic metastasis. Am. J. Clin. Pathol., 118: 425-434.
Direct Link - Graff, J.R., E. Gabrielson, H. Fujii, S.B. Baylin and J.G. Herman, 2000. Methylation patterns of the E-cadherin 5CpG island are unstable and reflect the dynamic, heterogeneous loss of E-cadherin expression during metastatic progression. J. Biol. Chem., 275: 2727-2732.
Direct Link - Gumbiner, B.M., 2000. Regulation of cadherin adhesive activity. J. Cell. Biol., 148: 399-404.
Direct Link - Guriec, N., L. Marcellin, B. Gairard, H. Calderoli and A. Wilk et al., 1996. E-cadherin m RNA expression in breast carcinomas correlates with overall and disease-free survival. Invasion Metastasis, 16: 19-26.
Direct Link - Handschuh, G., S. Candidus, B. Luber, U. Reich and C. Schott et al., 1999. Tumour-associated E-cadherin mutations alter cellular morphology, decrease cellular adhesion and increase cellular motility. Oncogene, 18: 4301-4312.
PubMedDirect Link - Heimann, R., F. Lan, R. McBirde and S. Hellman, 2000. Separating favorable from unfavorable prognostic markers in breast cancer: The role of E-cadherin. Cancer Res., 60: 298-304.
PubMedDirect Link - Hunt, N.C.A., A.G. Douglas-Jones, B. Jasani, J.M. Morgan and M. Pignatelli, 1997. Loss of E-cadherin expression associated with lymph node metastasis in small breast carcinomas. Virchows Arch, 430: 285-289.
CrossRefDirect Link - Jamal, A., R. Siegel, E.L. Ward, T. Murray, J. Xu, M. Smigal and J. Thun, 2006. Cancer Statistics. CA Cancer J. Clin., 56: 106-130.
CrossRefDirect Link - Kanai, Y., T. Oda, H. Tsuda, A. Ochiai and S. Hirohashi, 1994. Point mutation of the E-cadherin gene in invasive lobular carcinoma of the breast. Jpn. J. Cancer Res., 85: 1035-1039.
PubMed - Kleer, C.G., K.L. van Golen, T. Braun and S.D. Merajver, 2001. Persistent E-cadherin expression in inflammatory breast cancer. Mod. Pathol., 14: 458-464.
Direct Link - Larue, L., M. Ohsugi, J. Hirchenhain and R. Kemler, 1994. E-cadherin null mutant embryos fail to form a trophectoderm epithelium. Proc. Natl. Acad. Sci. USA., 91: 8263-8267.
Direct Link - Nass, S.J., J.G. Herman, E. Gabrielson, P.W. Iversen, F.F. Parl, N.E. Davidson and J.R. Graff, 2000. Aberrant methylation of the estrogen receptor and E-cadherin 5 CpG islands increases with malignant progression in human breast cancer. Cancer Res., 60: 4346-4348.
Direct Link - O'Shaughnessy, J.A., 2006. Molecular signatures predict outcomes of breast cancer. Eng. J. Med., 355: 615-617.
Direct Link - Rashid, M.G., M.G. Sanda, C.J. Vallorosi, J. Rios-Doria, M.A. Rubin and M.L. Day, 2001. Posttranslational truncation and inactivation of human E-cadherin distinguishes prostate cancer from matched normal prostate. Cancer Res., 61: 489-492.
Direct Link - Rubin, M.A., N.R. Mucci, J. Figurski, A. Fecko, K.J. Pienta and M.L. Day, 2001. E-cadherin expression in prostate cancer: A broad survey using high-density tissue microarray technology. Hum. Pathol., 32: 690-697.
Direct Link - Sarrio, D., G. Moreno-Bueno, D. Hardisson, C. Sanchez-Estevez, M. Guo, J.G. Herman, C. Gamallo and M. Esteller J. Palacios, 2003. Epigenetic and genetic alterations of APC and CDH1 genes in lobular breast cancer: Relationships with abnormal E-cadherin and catenin expression and microsatellite instability. Int. J., Cancer, 106: 208-215.
CrossRefDirect Link - Siitonen, S.M., J.T. Kononen, H.J. Helin, I.S. Rantala, K.A. Holli and J.J. Isola, 1996. Reduced E-cadherin expression is associated with invasiveness and unfavorable prognosis in breast cancer. Am. J. Clin. Pathol., 105: 394-402.
PubMed - Singletary, S.E., C. Allred, P. Ashley, L.W. Bassett and D. Berry et al., 2002. Revision of the American joint committee on cancer staging system for breast cancer. J. Clin. Oncol., 20: 3628-3636.
CrossRefDirect Link - Vos, C.B., A.M. Cleton-Jansen, G. Berx, W.J. de Leeuw and N.T. Ter Haar et al., 1997. E-cadherin inactivation in lobular carcinoma in situ of the breast: An early event in tumorigenesis. Br. J. Cancer, 76: 1131-1133.
PubMed - Wahed, A., J. Connelly and T. Reese, 2002. E-cadherin expression in pleomorphic lobular carcinomas: An aid to differentiation from ductal carcinomas. Ann. Diagn Pathol., 6: 349-351.
Direct Link - Woodward, W.A., E.A. Strom, S.L. Tucker, M.D. McNeese and G.H. Perkins et al., 2003. Changes in the 2003 America joint committee on cancer staging system for breast cancer dramatically affect stage-specific survival. J. Clin. Oncol., 21: 3244-3248.
Direct Link








