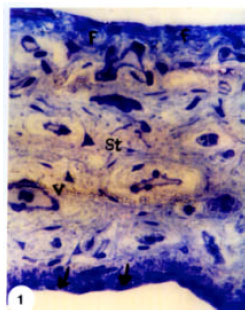
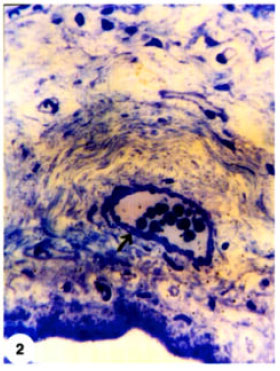
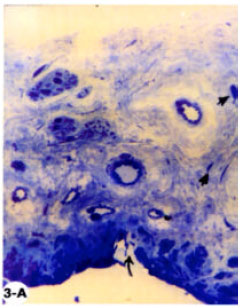
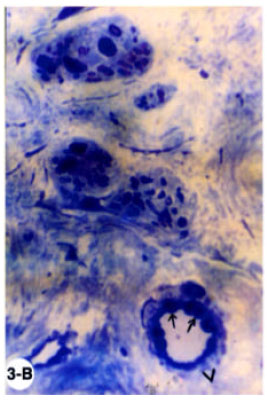
Research Article
Structural Changes in Rabbit Iris Following Excimer Laser Treatment
M.D.F.R.C.S. Ophthalmology Department, Research Institute Ophthalmology, 732 Cairo, Egypt
Mohamed A. El-Malt
M.D.F.R.C.S. Ophthalmology Department, Research Institute Ophthalmology, 732 Cairo, Egypt
Sawsan Karam
Pathology Department, Research Institute Ophthalmology, 732 Cairo, Egypt
Heba Issa
Pathology Department, Research Institute Ophthalmology, 732 Cairo, Egypt