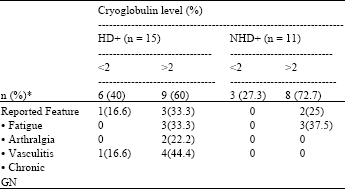
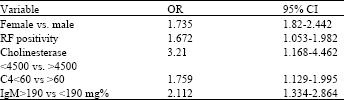
Research Article
Clinical Spectrum of Hepatitis-Associated Cryoglobulinemia: Cross-Link between Hematological and Immunological Phenomena
Department of Hematology, Theodor Bilharz Research Institute, Egypt
Nadia Amin Hussein
Department of Hematology, Theodor Bilharz Research Institute, Egypt
Lobna Yousef Ghanem
Department of Electron Microscopy, Theodor Bilharz Research Institute, Egypt
Abeya Abdel Megeed Lotfy
Department of Electron Microscopy, Theodor Bilharz Research Institute, Egypt
Manar Abdel Raouf Raafat
Department of Nephrology, Theodor Bilharz Research Institute, Egypt