Research Article
The Comparison of Misoprostol and Dinoprostone for Termination of Second Trimester Pregnancy
Department of Obstetrics and Gynecology, Department of Anesthesia, Zahedan University of Medical Sciences, Zahedan, Iran
Options for performing terminations of pregnancy in the second trimester include dilation and evacuation (D and E) and medical induction of labor. D and E require specialized equipments and training and has some complications such as cervical lacerations, uterine perforation and injury to abdominal viscera (Maitre et al., 2000; Creinin, 2000; Jain and Mishell, 1994). In contrast, almost all programs that provide abortion training offer medical abortion with prostaglandins (Pongsatha and Tongsong, 2003). Prostaglandines compounds either alone or in combination with other abortifacient agents has been used for termination of pregnancies in second trimester due to congenital fetal anomalies and intrauterine fetal demise. Intravaginal dinoprostone, the prostaglandin E2, has been widely used for second trimester pregnancy termination and has been shown to be safe and effective (Owen et al., 1992). Cost of the dinoprostone is one of the major concerns of gynecologists. Besides the cost, it must be stored in a refrigerator.
Misoprostol, a synthetic prostaglandin E1 analog is widely prescribed for the prevention and treatment of peptic ulcers. Also misoprostol has been commonly used for induction of labor at term (Rouzi, 2003; Herabutya et al., 2003) and termination in first and second trimester pregnancies (Daskalakis et al., 2005; Dickinson, 2005; Zieman et al., 1997; Jannet et al., 1996; Herabutya et al., 2001; Dickinson and Evans 2002; Pongsatha and Tongsong, 2004; Creinin et al., 2005). Compared with other prostaglandin preparations, misoprostol is cheaper and easier to use. It is stable at room temperature, readily available and quite inexpensive. Misoprostol can be administered orally, vaginally, or by other routes. Compared with the oral route, vaginal administration of misoprostol for second-trimester pregnancy termination results in greater efficacy and the side effects were also less frequent (Nigam et al., 2006; Eng and Guan, 1997; Chen et al., 1999; El-Refaey et al., 1995). After vaginal administration of misoprostol, plasma concentration gradually increased, reaching maximum levels after 70-80 min and slowly declining with detectable levels present beyond the 6 h study period (Tang and Ho, 2006). The optimal dosing regimen has yet to be determined. Studies have used doses ranging from 200 to 800 μg at intervals ranging from 3 to 12 h (Daskalakis et al., 2005). However, it has been reported that use of misoprostol for second trimester termination of pregnancy is associated with uterine rupture, especially when combined with oxytocin infusion (Costa and Vessey, 1993).
In this study we compared vaginal misoprostol and dinoprostone for termination of second trimester pregnancies due to intrauterine fetal death.
This clinical study was performed in Ali-ebne-abitaleb Hospital from July 2003 to July 2004. Forty women with singleton pregnancy at 13 to 27 weeks gestation and normal physical examination underwent for second trimester termination due to intrauterine fetal demise were included in this study.
The present prospectively designed study was approved by the ethics and clinical studies committee of Zahedan University of medical sciences and informed and signed consent was obtained from all the patients who were enrolled in the study.
Exclusion criteria included multiple gestation, placenta previa and primary use of a medication other than misoprostol and history of evidence of thromboembolism, Patients who had proven Prostaglandin allergy or any major health problem, smoking >10 cigarettes daily and breast feeding and presence of intrauterine contraceptive device. Accurate assessment of gestational age was established by reliable menstrual history and confirmed by ultrasonography in first trimester. The diagnosis of fetal demise was certain by ultrasonography.
Patients divided to two equal groups. Patients in group 1 (n = 20) received misoprostol 400 μg vaginally. Initial dose was repeated every 12 h (not to exceed 4 doses), unless labor was initiated and maintained regularly. In group 2 (n = 20) patients received dinoprostone gel 0.5 mg vaginally. If contractions of uterine were not started, oxytocin infusion was began 3 h after the initial dose; followed by an hour of crystalloid solution with no oxytocin, oxytocin initiated at 4 m units/min and increased by 1-2 m units/min increments every 15 min depending on uterine response (Mendilcioglu et al., 2002).
We collected information on maternal age, parity, gestational age, previous cesarean section, previous abortion. Following prostaglandin administration, pulse rate, blood pressure, temperature, systemic symptoms were monitored and recorded hourly. When the conception products were expelled, the patient's were evaluated by ultrasound for the presence of conception products still remaining in the uterine cavity. If there were no remnants (or minimal tissue was present), the abortion was considered complete and no further action was undertaken. Oxytocin infusion was administered for all women after delivery. The time interval between the first administrations of prostaglandin to delivery was assessed. Also proportion of subjects delivered vaginally by 24, 48 h and during the same admission was assessed.
The side effects and the need for operation delivery of placenta and the amount of blood loss during delivery were also assessed. The mean dose of oxytocin using in each group also were recorded. Uterine tachysystole was defined as six or more contractions in any 10 min periods and if occurred was recorded.
Data were analyzed using the SPSS 10 for Windows. The unpaired Student t-test or wilkoxone rank sum was used for continuous data. Categorical data were analyzed with the chi-square and Fisher exact tests. All tests of statistical significance were 2-tailed and p<0.05 was considered significant.
Twenty of 40 women enrolled in the study received vaginal misoprostol and 20 of them received vaginal dinoprostone. There were no significant differences among for demographic and historic characteristics of two groups such as age, gestation at delivery, gravidity, abortion, birth weight (Table 1).
In the misoprostol group mean gestational age was 19.8 week and after initial administration of 400 μg of misoprostol vaginally, 5 (25%) aborted within 6 h. All of the remainder of cases aborted within 24 h.
In the dinoprostone group mean gestational age was 20.1 week and after initial administration of dinoprostone gel 0.5 mg vaginally, 1(5%) aborted within 6 h. All of the remainder of cases aborted within 24 h.
The mean induction delivery time interval was 13.2 h in the vaginal misoprostol group and 15.1 h in the vaginal dinoprostone group and there was no significant differences for the mean induction delivery time interval between groups (Table 2).
The frequency of PG administration were not with significant differences between groups.
The mean dose of oxytocin usage was 50.3±15.3 units in misoprostol group and 146±14.5 units in the dinoprostone group and there was significant differences between groups (p = 0.01).
In patients with a previous vaginal birth, time to delivery was twice as short as patients without a previous vaginal birth (HRR 1.9 95% CI 1.2-2.9) (Table 2).
| Table 1: | Demographic characteristics of women in groups |
 | |
| Data are presented as % or mean±SD | |
| Table 2: | Comparison of outcomes in two groups |
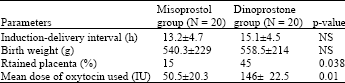 | |
| Data are presented as % or mean±SD | |
Nausea was seen in 8 (40%) of patients in misoprostol group and 4 (20%) of patients in dinoprostone group and vomiting was seen in 6 (30%) of patients in misoprostol group and 3 (15%) of patients in dinoprostone group. Uterine tachysystole and uterine rupture did not occur in any women in two groups, also no other major complication was observed among the patients and treatment was not stopped in any of the patients (Table 2). Operative placenta removal occurred in 15% of the women in the misoprostol group versus 45% in the dinoprostone group (p = 0.038).
The median amount of blood loss was 150 mL in both groups and no blood transfusion required in any of the study groups.
Time to delivery is commonly used in studies for measuring effectiveness for induction of labor. This study confirms the efficacy of misoprostol compare to dinoprostone for termination of second trimester.
In this study the mean induction delivery time interval was 13.2 h in the vaginal misoprostol group and 15.1 h in the vaginal dinoprostone group and there was no significant difference between groups.
Jain and Mishel (1994) could not find any significant difference between the vaginal dinoprostone and misoprostol regimens for termination of pregnancies between 12 and 22 weeks (Jain and Mishell, 1994). These results were similar to our study and other studies (Mendilcioglu et al., 2002; Roel et al., 2004), although Jain and Mishel (1999) could used a higher dose of dinoprostone and lower dose of misoprostol.
In Mendilcioglu (2002) study using a combination of 400 μg oral with 600 μg vaginal misoprostol compared with 600 μg vaginal misoprostol and also compared with 0.5 mg dinoprostone gel, a mean induction delivery time interval of 20.3 h for oral vaginal and 17.3 h for vaginal misoprostol and 22.5 h for dinoprostone was achieved. There was no significant difference between groups in their study.
Also as reported by Roel de Heus et al. (2004), mean induction delivery time interval was 16.5 h in women with fetal death using misoprostol. The mean induction delivery time interval in our study was shorter than these studies. In Heus's study misoprostol dosage regimen was 100 μg every 12 h. In this study misoprostol dosage was 400 μg every 12 h.
In other studies mean induction delivery time reported to be 12.6-14.8 h (Eng and Guan, 1997; Bugalho et al., 1994; Merrell and Koch, 1995), these results were similar to present study.
Bugalho et al. (1994) found a median time to delivery as 12.6 h in their study (n = 72) and a delivery rate of 92% within 24 h. Misoprostol dosage regimen was 100 μg every 12 h, but mean gestational age at induction was 31.2 weeks, as compared to 20 weeks in this study.
An optimal dose of misoprostol leading to a significant diminished time to delivery interval without an increase in side effects and possible complications for terminations of pregnancy in the second trimester has not yet been found.
A single dose of 200 μg of vaginal misoprostol was found to be sufficient to induce second-trimester abortion in most patients (Bugalho et al., 1993). However, it was reported that higher doses yielded shorter induction-delivery intervals than 200 μg of misoprostol which offer less hospitalization and better patient tolerance to induction (Dickinson and Evans, 2002). Doses of 600 and 800 μg of misoprostol have been evaluated for termination of pregnancy in the second trimester. However, these doses are associated with unacceptably high rates of fever, diarrhea nausea and vomiting (Herabutya et al., 2000; Pongsatha and Tongsong, 2001).
In this study, with using a 400 μg vaginal misoprostol a mean induction delivery time of 13.2 was achieved, but the regimen using 400 μg every 6 h effected delivery more rapidly (Rodney et al., 2005).
Eng and Guan (1997), by using intravaginal misoprostol, found that the abortion rate was influenced by the gestational age; they reported 100% abortion rate for patients>17 weeks' gestation, compared to 67% for those with a gestational age of 13-16 weeks. In this study 100% of cases aborted within 24 h.
Some studies demonstrated that time to delivery is shorter in women with fetal death as compared to viable pregnancies and advise in viable pregnancies the use of misoprostol in combination with mifepristone (Roel de Heus et al., 2004).
Mendilcioglu et al. (2002) reported in their study that the requirement of oxytocin for induction of labor was higher in the dinoprostone group than misoprostol group. This finding is similar to the result of this study for the oxytocin's requirement in two groups.
Induction of labour with misoprostol in women with a previous cesarean delivery still remains a matter of concern. Several reports describe uterine rupture associated with misoprostol or sulprostone and tachysystole (De Boer et al., 2001; Berghahn et al., 2001; Wing et al., 1998; Prasad and Ratnam, 1992; Prasad and Ratnam, 1992). In this study uterine rupture and tachysystole was not occurred in any patients with and without previous cesarean delivery.
Some studies describe nausea and vomiting as most common side effects for both misoprostol and dinoprostone (De Boer et al., 2001; Jain et al., 1999; Herabutya et al., 2001). Side effects are dose dependent and decreased when misoprostol is administered vaginally (Herabutya et al., 2001).
Also in present study the most common side effects for both misoprostol and dinoprostone were nausea and vomiting and no other major complication was observed.
In conclusion based on present results, no statistical difference was observed in terms of effectiveness of both the misoprostol and dinoprostone regimens and both regimens are quite safe, but misoprostol is a cost-effective method for termination of pregnancy and can be stored at room temperature.
Further analysis are needed for optimization of misoprostol dosing schedules in second trimester pregnancy termination. Also further studies on combination therapy, complications and patients preferences are warranted.