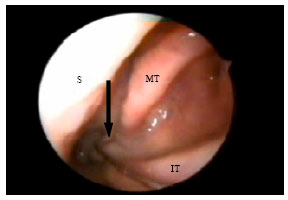
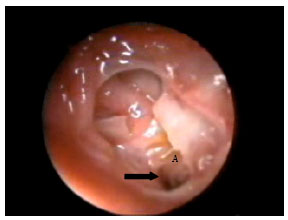
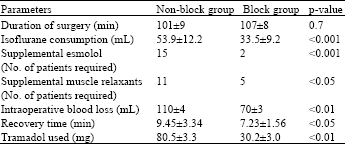
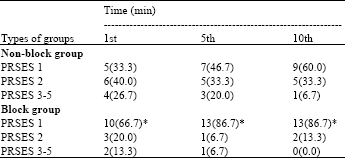
Research Article
Role of Intraoperative Endoscopic Sphenopalatine Ganglion Block in Sinonasal Surgery
Department of Anesthesia and ENT Surgery, Faculty of Medicine, Asian Network for Scientific Information Cairo University, Egypt
Md. Ehab H. Abu-Zaid
Department of Anesthesia and ENT Surgery, Faculty of Medicine, Asian Network for Scientific Information Cairo University, Egypt