Research Article
The Effects of Carbenoxolone on the Contractile Responses of the Isolated Guinea-Pig and Rat Smooth Muscles
Department of Pharmacology, Qazvin University of Medical Sciences, Qazviru P.O Box 341197-5981, Islamic Republic of Iran
INTRODUCTION
Glycyrrhiza glabra is a perennial herb in Iran and it has several traditional medicinal properties and pharmacological activities which include the treatment of peptic ulcers, asthma, pharyngitis and abdominal pain (Blumenthal et al., 2001; Leung, 1980). It has been shown expectorant, antitussive, antidote, anti-inflammatory, antioxidant activity and hepatoprotective properties (Moon and Kim, 1997; Tangri et al., 1965; Kiso et al., 1984; Haragushi et al., 1998). Bioactive components of licorice root are the triterpene glycoside, glycyrrhizic acid (GL) and its aglycone, 18b-glycyrrhetinic acid (GLA) (Blumenthal et al., 2001). GL is a noteworthy drug among phythotherapeutics and its chemical modification is the perspective route to design new bioactive compounds for medicine. Carbenoxolone is now used in clinical treatment of ulcer diseases and has some pharmacological properties such as the inhibition of gap junctional intercellular communication (Davidson and Baumgarte, 1988; Turpie and Thomson, 1965). Gap junctions are believed to be located at regions of close apposition of the surface membranes of neighboring cells, in the form of intercellular ion channels connecting their interiors (Lowenstein, 1981). The contractile response of mammalian smooth muscle may depend upon the spread of excitation through gap junctions (Bennet, 1973). It has been suggested that the cellular network of interstitial cells of Cajal (ICC) connected by gap junctions, plays an important role in the regulation of gut motility (Sanders, 1996). Thus the aim of the present study was to study the effects of carbenoxolone to the contractile responses of the isolated colon and the isolated guinea-pig ileum to different stimuli.
Animals: Male Wistar rats (250-300 g) and guinea-pigs (300-350 g) were obtained from the Razi Institutes, Mashhad, Iran. The animals were housed in colony rooms with 12/12 h light/dark cycle at 21±2°C and had free access to food and water. All animal experiments were carried out in accordance with Mashhad University of Medical Sciences, Ethical Committee Acts.
Chemicals: Carbenoxolone was obtained from Sigma. Acetylcholine chloride was purchased from Merck. Sodium thiopental (1 g) was obtained from Biochemie (GmbH, Vienna-Austria). All drugs were dissolved in distilled water.
Isolated rat colon: Rats were anaesthetized with sodium thiopental (60 mg kg-1 by i.p. injection). The segments of proximal colon (2 cm) were removed and rinsed of intraluminal content and full-thickness preparations of them were used. The strips were suspended vertically in 50 mL organ baths for isometric recording of mechanical activity using a Physiograph (Ossillograph 400 MD/2). An initial load of 1 g was applied to the all segments, the Krebs solution being changed every 15 min. The Krebs solution had the following composition (mM): NaCl 118.4, KCl 4.7, MgSO4 7H2O 1.4, KH2PO4 1.2, CaCl2 2.5, NaHCO3 25 and glucose 11.1.
Isolated guinea-pig ileum: Guinea-pigs were killed by a blow to the head and exsanguinations. The ileum was exteriorized, the ileo-caecal junction located and approximately 30 cm removed. A 10 cm segment of the terminal portion was discarded before the contents in the remaining ileum were flushed out with Krebs solution of the same composition as mentioned above. An initial load of 1 g was applied to the full-thickness preparations of the ileum (2 cm), the Krebs solution being changed every 15 min then ready for isometric recording of mechanical activity. In all experiments, concentration response curves to carbenoxolone were established using a cumulative protocol: each muscle strip was exposed to several concentrations of carbenoxolone until the response reached its maximum.
Electrical field stimulation: Electrical field stimulation was provided by a Grass S88 stimulator. EFS was applied using a ring electrode applying supramaximal trains of pulses (10 Hz, 0.3 ms pulses) for ileum and colon segments. A voltage greater than which produced a maximal twitch has been applied in both experiments.
Analysis of data: Values in the text refer to mean±SEM. Differences between groups have been compared using One Way ANOVA followed by a Tukey-Kramer multiple comparison test.
Effects on the isolated rat colon: EFS induced contractions in the isolated rat colon. Preincubation of the tissue with carbenoxolone (contact time 90 min) significantly decreased the contractility induced by EFS (Fig. 1A) and by acetylcholine (0.1 μM) in a concentration-dependent manner (Fig. 1B). Carbenoxolone (0.05-5 μM) reversibly reduced Spontaneous Mechanical Activity (SMA) of the isolated rat colon (Fig. 2).
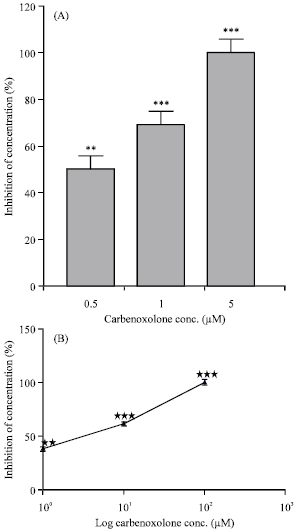 | |
| Fig. 1: | Inhibition of contractions induced by electrical field stimulation (A) and by 0.1 μM acetylcholine (B) in the isolated rat colon by the carbenoxolone. Each column or point represents the mean±SEM of inhibition expressed as percentage of the amplitude of control contractions (n = 4). **p<0.01, ***p<0.001, Tukey-Kramer Test |
Effects on the isolated guinea-pig ileum: Preincubation of the tissue with carbenoxolone (contact time 90 min) significantly decreased the contractility induced by EFS (Fig. 3A) and by 1 μM acetylcholine (Fig. 3B) in a concentration-dependent manner. Carbenoxolone (0.1-10 μM) reduced SMA of the isolated guinea pig ileum (Fig. 4).
In order to determine whether any possible action of carbenoxolone on smooth muscle contractility was tissue or species-dependent, two different preparations including the isolated rat colon and the isolated guinea-pig ileum were used.
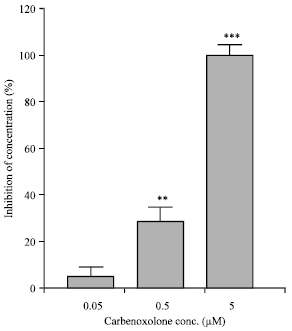 | |
| Fig. 2: | The inhibitory effect of carbenoxolone (0.05-5 μM) on spontaneous mechanical activities of the isolated rat proximal colon. Each column represents the mean±SEM of inhibition expressed as percentage of the amplitude of control contractions (n = 4). **p<0.01, ***p<0.001, Tukey-Kramer Test |
The responses evoked by EFS in the isolated rat colon and the isolated guinea-pig ileum are mainly induced by muscarinic receptors activation. In this study carbenoxolone also decreased SMA and acetylcholine-induced contraction in the isolated rat colon and in the isolated guinea-pig ileum. Gap junctions may conduct neural message between ICCs of myenteric plexus (ICC-MP) (Daniel and Wang, 1999; Daniel et al., 1998). Few gap junctions exist in the mouse intestine between ICC-MP either longitudinal or circular muscles (Thuneberg, 1982; Daniel and Wang, 1999), but in the rat intestine these channels have been found between ICC-MP and muscles (Komuro, 1985). Also, it was identified two networks of ICC associated with submuscular (ICC-SMP) and Auerbach’s (ICC-AP) plexuses in the rat colon and ICCs at the submucosal border are responsible for high frequency (HF) contractions the rat colon (Pluja et al., 2001; Alberti et al., 2005). Gap junctions are found in the submuscular area of the colon and deep muscular plexus of the canine small intestine. They also have been observed between circular muscle cells. But longitudinal muscle cells have these channels only near the myenteric plexus of colon. Some electrical couplings between them and circular muscles have been observed (Daniel et al., 2001). Also, in another study was observed dye coupling among ICC-MP in guinea-pig small intestine (Bayer, 2001).
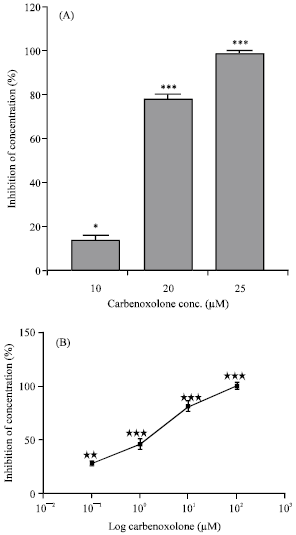 | |
| Fig. 3: | Inhibition of contractions induced by electrical field stimulation (A) and by 0.1 μM acetylcholine (B) in the isolated guinea-pig ileum by the carbenoxolone. Each column or point represents the mean±SEM of inhibition expressed as percentage of the amplitude of control contractions (n = 4). *p<0.05, **p<0.01, ***p<0.001, Tukey-Kramer Test |
SMA is a result of spontaneous firing of a number of cholinergic neurons (Belzer et al., 2002). Carbenoxolone inhibited the actions of the mediators at concentrations similar lower to those required inhibiting responses to EFS in every case. The result that carbenoxolone inhibited the agonist-induced contraction at concentration that was lower to inhibit EFS-induced one, it does not inhibit a gap junction suggests that it depresses directly the smooth muscle contraction. Thus, the inhibitory effects of carbenoxolone on contractile response of muscles may be related to other effects of it. There is some evidence that glycyrrhetinic acid derivatives exerted a variety of effects such as alter the activity of ion transport processes, including ion channels and inhibition Na+-K+-ATPase (Terasawa et al., 1992; Tare et al., 2002).
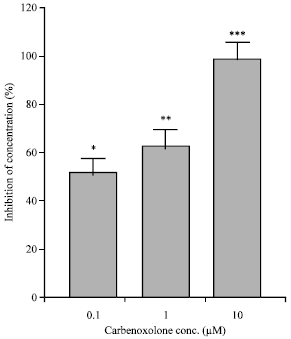 | |
| Fig. 4: | Inhibitory effect of carbenoxolone on spontaneous mechanical activities of the isolated guinea-pig ileum. Each column represents the mean±SEM of inhibition expressed as percentage of the amplitude of control contractions (n = 4). *p<0.05, **p<0.01, ***p<0.001, Tukey-Kramer Test |
It was reported that glycyrrhetinic acid raised [Ca2+]I in single cell which was unrelated to block gap junctions (Jonkers et al., 1999). In contrast to above finding it was also demonstrated that carbenoxolone is a selective uncoupling agent that does not affect ion currents (de Groot et al., 2003). On the other hands, there may be other excitable system exist independent of ICC-MP in the intestine which is responsible to explain these effects which was described in previous study (Daniel, 2004). Recently similar results have shown that carbenoxolone didn’t change IJPs in the smooth muscle of mouse proximal colon (Sibaev et al., 2006). It was also suggested that there is no evidence that gap junction blockers are involved in neuromuscular transmission in mouse colon (Sibaev et al., 2006).
Carbenoxolone appears to inhibit both tissues contractility and agonist efficacy subsequent to activation of cholinergic receptors which seems unrelated to uncoupling effects of it in the isolated rat colon and the isolated guinea-pig ileum. Further studies are still required to confirm unequivocally the mechanisms by which carbenoxolone inhibits contractile responses in these tissues.