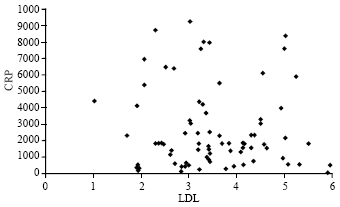
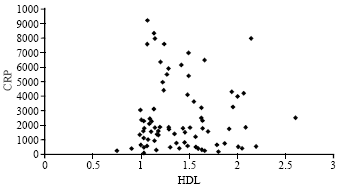
Research Article
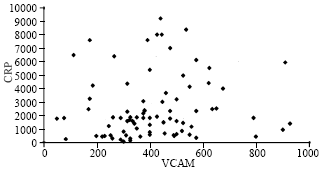
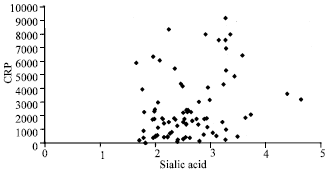
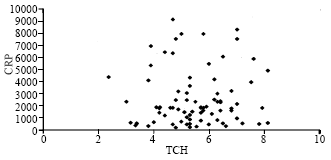
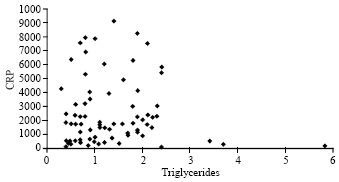
Correlation of Plasma C-reactive Protein Levels to Sialic Acid and Lipid Concentrations in the Normal Population
Department of Medicine, Faculty of Medicine, University of Malaya, Kuala Lumpur, Malaysia
Ikram Shah Ismail
Department of Medicine, Faculty of Medicine, University of Malaya, Kuala Lumpur, Malaysia
Sekaran Muniandy
Department of Molecular Medicine, Faculty of Medicine, University of Malaya, Kuala Lumpur, Malaysia
Kumutha Malar Vellasamy
Department of Medicine, Faculty of Medicine, University of Malaya, Kuala Lumpur, Malaysia
Karuthan Chinna
Faculty of Information Technology and Quantitative Sciences, MARA University of Technology, Shah Alam, Selangor, Malaysia