Research Article
Antioxidant Activity and Inhibition of Human Cancer Cells by the Herbal Product, ARCOMIG
Molecular Biology and Genetics, 245 Biotechnology Bldg., Cornell University, Ithaca, NY 14853-2703, USA
John P. Berry
Division of Marine Biology and Fisheries, Rosenstiel School of Marine and Atmospheric Science, University of Miami, Miami, FL, USA
Linson Cadena
Arcomig Natural Herbs and Teas, Huntington Park, CA, USA









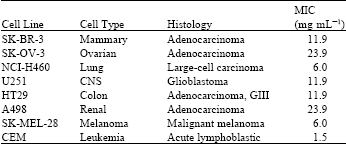
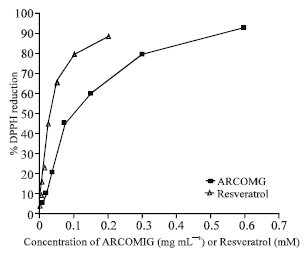
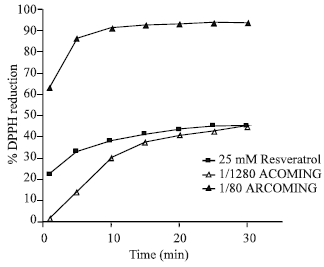
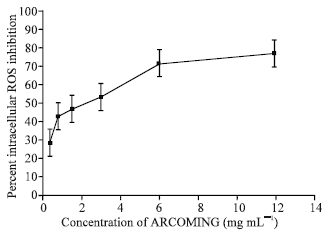
Estella Reply
Good afternoon!
Where in the USA can I purchase your product.