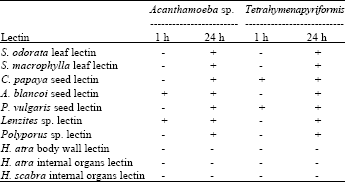
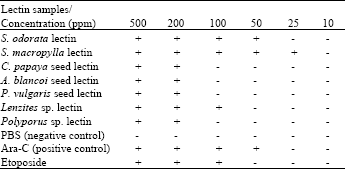
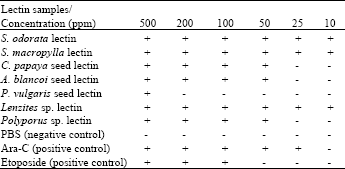
Research Article
Evaluation of Some Lectins as Anti-protozoal Agents
lnstitute of Biology, University of the Philippines, Diliman- Quezon City, Philippines
Elmer-Rico E. Mojica
Institute of Chemistry, University of the Philippines, Los Ba�os, Laguna, Philippines
Florinia E. Merca
Institute of Chemistry, University of the Philippines, Los Ba�os, Laguna, Philippines
Marivic S. Lacsamana
Institute of Chemistry, University of the Philippines, Los Ba�os, Laguna, Philippines
Custer C. Deocaris
Gene Function Research Center, I-I-I Higashi, AIST Central 4: Tsukuba 305-8562, Japan