Research Article
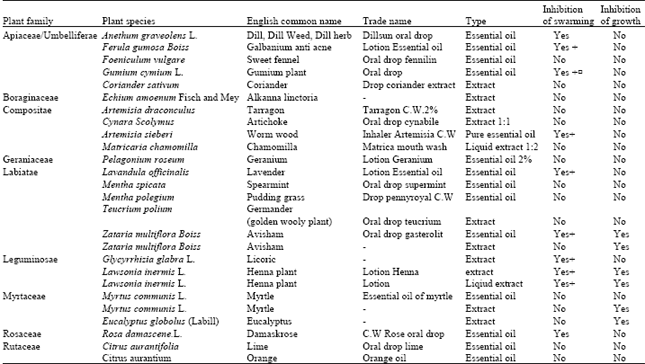
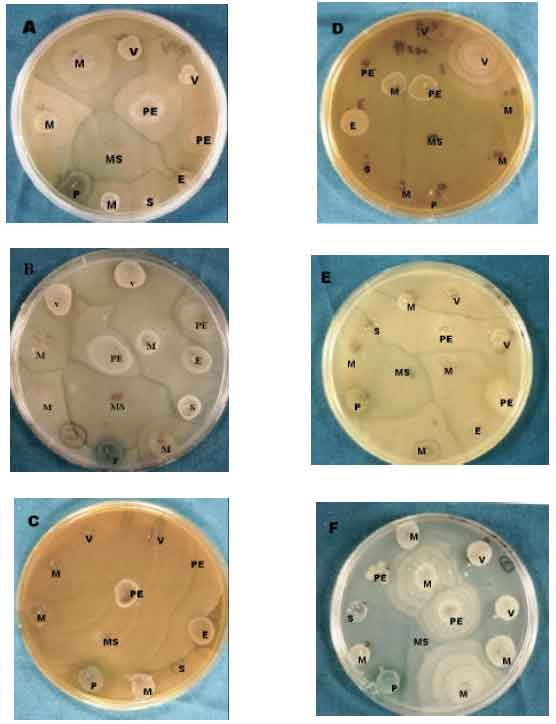
Inhibitory Effect of Some Medicinal Plants from Iran on Swarming Motility of Proteus Rods
Department of Microbiology Kerman University of Medical Sciences P.O. Box 444 Kerman, Iran
Afshin Amari
Department of Microbiology Kerman University of Medical Sciences P.O. Box 444 Kerman, Iran
Ali Ghanberi Asad
Department of Microbiology Kerman University of Medical Sciences P.O. Box 444 Kerman, Iran