Research Article
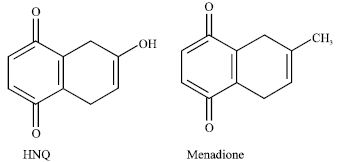
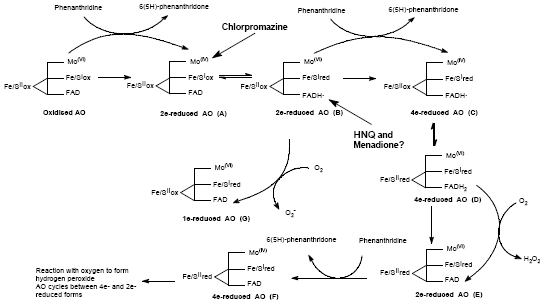
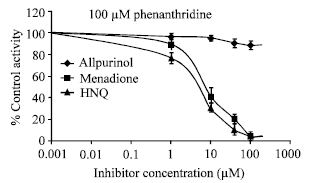
Effect of 2-hydroxy-1, 4-naphthoquinone, a Natural Dye of Henna, on Aldehyde Oxidase Activity in Guinea Pig Liver
Department of Pharmaceutical Chemistry College of Pharmacy King Saud University P.O. Box 2457, Riyadh 11451 Saudi Arabia