Research Article
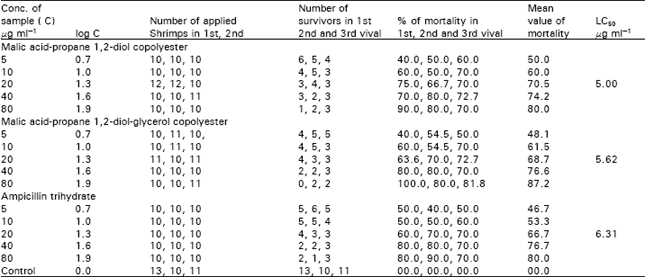
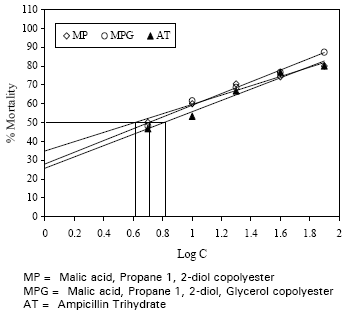
Toxicological Study of Malic Acid-Propane 1, 2-Diol and Malic Acid-Propane 1, 2-Diol-Glycerol Copolyesters on Brine Shrimp Nauplii
Department of Applied Chemistry and Chemical Technology, University of Rajshahi, Rajshahi-6205, Bangladesh
M. Ahmed
Department of Pharmacy, University of Rajshahi, Rajshahi-6205, Bangladesh
M. A. Islam
Department of Applied Chemistry and Chemical Technology, University of Rajshahi, Rajshahi-6205, Bangladesh
M. R. Karim
Department of Applied Chemistry and Chemical Technology, University of Rajshahi, Rajshahi-6205, Bangladesh
M. H. U. Biswas
Department of Applied Chemistry and Chemical Technology, University of Rajshahi, Rajshahi-6205, Bangladesh