Research Article
Kinetics Parameters of Na+, K+-ATPase from Different Mice Brain Lobes
Department of Biochemistry, Faculty of Science, King Abdulaziz University, P. O. Box- 80203, Jeddah-21 589, Saudi Arabia
The surface membrane of practically all animal cells is studded with millions of copies of Na+, K+-ATPase. Extrusion of cellular Na ions in exchange for intracellular K ions, at the expense of a major fraction of the cell’s ATP budget, is responsible for maintaining the steep concentration gradients of these ions, inwardly directed for Na and outwardly for K, that are vital to the life of the cell. These gradients drive numerous co- and counter-transporters that supply the cells with glucose, amino acids and other building blocks, they regulate cell volume, pH and Ca homeostasis and they underlie nearly all the electrical activity in the peripheral and central nervous system and in cardiac and skeletal muscle (Beauge et al., 1997).
It is well known that Na+, K+-ATPase is involved in the restoration and maintenance of sodium and potassium equilibrium through neuronal membranes both at rest and after passage of a nervous impulse (Stahl, 1986; Wu, 1986). The Na+, K+-ATPase is a highly concentrated in synaptic nerves endings, and regulatory mechanisms that control this enzymatic activity are essential in maintaining metabolic activity of the synaptic region and in processes directly related to neurotransmission (Wu, 1986).
Na+, K+-ATPase is characterized by complex kinetic behavior reflected in abnormal substrate dependance and described as a curve with an intermediary plateau (Boldyrev, 1991). This feature does not depends on the source of the enzyme studied (Lopina et al., 1995), but is closely connected to interprotomer interaction of the enzyme and the modulating effect of ATP on the activity (Wyse-Angela et al., 1998).
There is a substantial motivation for learning how these ubiquitous molecular machines work because each of the human pumps is a target for either potent therapeutic drugs or disease- causing mutations (Ganea et al., 1999).
Therefore, the research work was conducted to investigate the kinetics of the Na+, K+-ATPase from the five different lobes of swiss albino mice brain and compared for Vmax and Km in order to obtain some information on related processes.
The Bovine serum albumin, Tris (Tris [hydroxymethyl] aminoethane), ATP (adenosine-5'-triphosphate, disodium salt, grade I), ascorbic acid, ouabain, EDTA (ethylenediamine tetra acetic acid, disodium salt, 2 H20) were obtained from Sigma Chemical Co., St. Louis, MO, USA. All other chemicals were of reagent grade. Male swiss albino mice (30-40 g) were used throughout this work.
Preparation of brain homogenate: Animals were adapted to laboratory atmosphere for two weeks under the same conditions. After the decapitation, brains were rapidly excised and frozen at -20°C, then dissected into the following lobes: Cerebral hemisphere I (Bl), Cerebral hemisphere II (B2), Thalamen cephalon (B3), Cerebellum (B4) and Medulla oblongata (B5) (Abdel - Raheem et al., 1979). The tissues were either worked upon immediately or stored at -20°C until the enzyme assay was started. Each brain lobe was accurately weighed then homogenized in 0.25M sucrose solution. The homogenate was diluted to give the proper enzyme activity which can be measured within a suitable absorbency range. Total protein concentration of each lobe was determined according to Lawry et al. (1951).
The phosphomolybdic assay was adopted to measure the number of “n” moles of inorganic phosphate released by the action of the ATPase as a measure of its activity according to the method of Serrano (1978). This method is based on selective inhibition of Na+, K+-ATPase by the glycoside ouabain. Accordingly, the ATPase activity was measured in the presence of ouabain to give Mg+2 ATPase activity and in its absence to give total ATPase activity and by subtraction, the Na+, K+-ATPase activity could be calculated. This reaction mixture was buffered with 50mM Tris pH 6.5 and contained different concentrations of MgCl2.6H2O, 100mM NaCl, 10mM KCl, 0.1mM EDTA and +/- 1.5μM ouabain. After adding the tissue homogenate (50μl) and incubation for 5min at 30°C in a water bath shaker, the reaction was started by the addition of different volumes of 0.1M ATP. After 10min incubation at 30°C, the reaction was stopped by the addition of 2ml of a solution containing 2% v v-1 sulfuric acid, 0.5% w v-1 ammonium molybdate and 0.5% w v-1 sodium lauryl sulfate. The detergent was included here to avoid the development of any turbidity. The phosphomolybdate was reduced with 20Fl of 10% w v-1 ascorbic acid and the absorbency at 750nm was read after 5min according to the method of Fiske and Subbarow (1925). Kinetic parameters (Km and Vmax) of Na+, K+-ATPase were calculated using a computer program (Stainslawski, 1996).
The data presented here is the result of triple experiments each consisting of 15 mice at least. Values given are mean ± standard errors (Norusis, 1989).
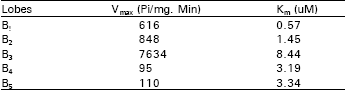
Fig. 1 shows the specific activity of Na+, K+-ATPase of different lobes from brain mouse at 2mM Mg-ATP. The Na+, K+-ATPase specific activity of lobe B3 has the highest activity followed by B2, Bl, B5 and B4 in decreasing order of activity. Table 1 represents the Michaelis-Menten (Km) values of Na+, K+ ATPase from different lobes. The maximum velocity (Vmax) of the enzyme from lobe B3 was the highest followed by B2, Bl, B5 and B4 in decreasing order of activity.
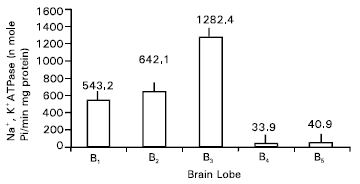 | |
| Fig. 1: | Specific activity of Na+, K+-ATPase from different lobes of swiss albino mice brain at 2mm Mg-ATP. |
| Table 1: | Kinetics parameters of Km and ( V max) of Na+, K+-ATPase from different albino mice brain |
 | |
 | |
The mammalian brain consists of several distinct parts, including the cerebrum, the cerebellum and the brainstem. The cerebrum is covered by the cerebral cortex, a layer of gray matter that houses the sites of higher brain functions. The cerebellum, in conjunction with the motor cortex of the cerebrum, controls complex motor activities. The brainstem controls involuntary visceral activities, such as breathing and heart rate and homeostatic functions (Brum et al., 1994).
Since in the brain, the Na+, K+-ATPase is the single most ATP consuming enzyme, being responsible for maintaining ion gradients (Hylland et al., 1997). Therefore, studying the activity of the Na+, K+ ATPase from different lobes of mice brain facilitates to kinetically characterize individual Na+, K+-ATPase isozymes. The information obtained from this investigation will give the opportunities to relate these kinetic parameters (Km, Vmax) with the roles that these isozyme plays in each lobe. This work demonstrate that different brain lobe has different Na+, K+-ATPase kinetics characteristics.
Km, the substrate concentration at which the reaction velocity is half-maximal showed different values for different lobes. In general, if an enzyme has a small value of Km, it achieves maximal catalytic efficiency at low substrate concentrations. Of course, the magnitude of Km varies widely with the identity of the enzyme and the nature of the substrate. It is also a function of temperature and pH (Voet and Voet, 1995).
The Na+, K+-ATPase maximal activity of lobe B3 has the highest activity followed by B2, Bl, B5 and B4 in decreasing order of activity. This may be suited for handling large Na+ loads in electrically active cells. This result is similar in general to the result of the experiment on sodium kinetics of Na+, K+-ATPase α isoforms in intact transfected HeLa cells (Zahler et al., 1997). Same conclusion was obtained by Pekovic (1997) in demonstrating striking differences in kinetic properties of the hippocampal and striatal Na, K-ATPase that may be due to the isoenzyme diversity and adaptations to specific physiological demands of the examined brain regions (Pekovic et al., 1997).
In addition, the data obtained from the work of Averet (1987) suggested that the same enzyme exhibit various responses due to dissimilar lipid composition of the Na, K-ATPase environment, and/or different adaptative responses to abnormal ion concentrations in different brain regions that could account for these different responses (Averet et al., 1987).