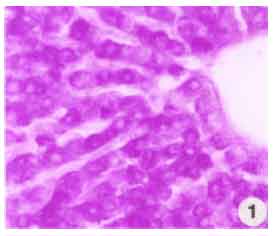
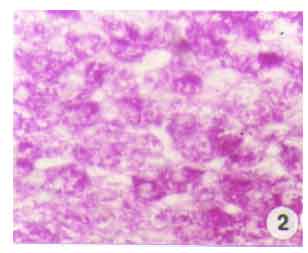
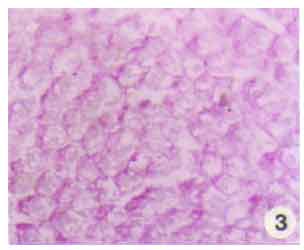
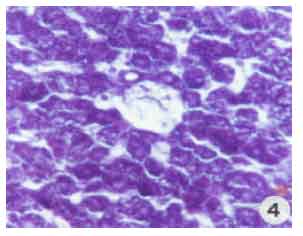
Research Article
Pyrethroid Inhalation Induced Histochemical Changes in the Liver of Albino Rats
Department of Zoology, Faculty of Science, Menoufia University, Egypt
F. A. El-Mesady
Department of Zoology, Faculty of Science, Menoufia University, Egypt
N. I. El-Desouki
Department of Zoology, Faculty of Science, Tanta University, Tanta, Egypt