Research Article
Cadmium Induced Toxicity on Pregnant Mice and Their Offspring: Protection by Magnesium or Vitamin E
Department of Zoology, Faculty of Science, Alexandria University, Alexandria, Egypt
Toxic metals have been shown to affect almost every organ system of the body, and often due to their poor handling and excretion, the chances of serious damage increase with continued exposure. It is well documented that heavy metals accumulate most highly in the mammalian liver and kidney, which are primary organs involved in heavy metal excretion. Extended exposure to heavy metals leads to chronic heavy metal intoxication in these organs (Markovich and James, 1999). An example of such toxic metals is cadmium (Cd+2). It is one of the most important environmental pollutants as a result of immense usage in plating industry, batteries, plastic and semiconductors (Friberg et al., 1986). Levels of Cd+2 in the soils and plants are increased by the application of phosphate fertilizers and sewage sludge. The more Cd+2 contaminates in the agricultural environment, the more it contaminates the food chain and endangers human consumers’ health, Thus it damage tissues and milk of grazing animals. Cd+2 is a non-biodegradable and has a very long biological half-life (10-30 years) in humans (Friberg et al., 1986).
Several estimates show that 50% of the provisional tolerate weekly intake of 400 to 500μg Cd+2 is still reached in various European countries and that this value can be exceeded by a factor of upto 2.5 in areas of extreme soil pollution (Elsenhans et al., 1997). Cadmium; is known to cause severe damage to various organs. “Itai-itai disease” was caused by long term exposure to Cd+2 in the Toyama Prefecture in Japan. Patients with this disease showed a nephropathy, osteomalacia and servers pain (Hamada et al., 1991). Anaemia is the major haematotoxic effect following long-term exposure to Cd+2 in humans (Horiguchi et al., 1994) and in laboratory animals (Hiratsuka et al., 1996).
Several metals have been shown to cause tolerance to Cd+2 – induced tissue injury (Zn+2, Cu+2 and Ni+2), steroid hormones (androgens, estrogens and progesterone), antioxidants (ascorbate and tocopherol), calmodulin inhibitors (chlorpromazine and trifluoperazine) and thiol containing compounds (cysteine and glutathione) ( Friberg et. al., 1986). The mechanism of tolerance varies among compounds, but in many cases involves the induction of metallothionein or direct chelation (Waalkes and Goering, 1990).
Magnesium has played an important role in the process of biological evolution towards more differentiated organisms with more effective energy utilization. Overall Mg+2 is important for >300 different enzyme systems (Wester, 1987).
Magnesium completely prevent the sharp increase in lung tumor formation in strain A mice induced by nickel and lead acetates (Poirier, et al., 1980). The results of Poirier et al. (1983) demonstrated clearly that the development of tumors at the site of s.c. cadmium chloride (CdCl2) injection can be prevented by simultaneous injection of magnesium acetate(CH3COO)2Mg at the same place. There is a good possibility that oxidative stress may be one of the mechanisms involved in Cd+2 toxicity. Shaikh et al. (1999) found that an increased in lipid peroxidation in both liver and kidney after Cd+2 administration. Protection against these actions of Cd+2 can be achieved through the antioxidant systems (Rana and Verma, 1996).
Vitamin E activity is derived from a series of dietary tocopherol and tocotrienols, the most potent of which is d-α-tocopherol. This vitamin serves as an antioxidant, protecting polyunsaturated fatty acids in membranes and possibly also in tissues from attack by free radicals (Wyngaarden and Smith, 1985).
Therefore, the present work aimed to study the toxic effects of Cd+2, administered during gestation, on pregnant mice and their offspring, as well as, the assessment of the potential protective effect of either Mg+2 or vit .E on the amelioration of Cd +2 induced toxic effects.
Materials and Methods
Cadmium: Pure cadmium chloride-1-hydrate (CdCl2.H2O) with a molecular weight of 210.33gm was used (Sigma Chem. Co.). It was dissolved in 0.9% saline solution.
Protective agents:
| (a) | Magnesium chloride (MgCl2) with a molecular weight of 46.94gm was utilized (Aldrich Chemical Co. Milwaukee, WI). It was dissolved in 0.9% saline solution. |
| (b) | Vitamin E (vit. E, α-tocopherol acetate) was purchased from Sigma Chemical Co (St. Louis, MO). It was dissolved in vegetable oil. |
All other chemicals were of highest purity commercially available.
Experimental animals and groups: Albino mice of Swiss strain were used as experimental animals. Ten males and females were allowed to breed in the laboratory in order to obtain inbred strain. Standard diet was provided and water was available ad libitum. Vaginal smears were examined and the different stages of the oestrus cycle were followed as described by Snell (1956). The females found in oesterous, were selected; each one was mated with one adult male from 8 to 10 p.m. The mid-point of the mating period was considered as the beginning of gestation. A total number of 120 pregnant mice were divided into 6 groups:
| Group (1): | The animals were subcutaneously injected with 0.1 ml of 0.9% saline solution for 10 successive days, starting from 3rd day of gestation. They served as a control group. |
| Group (2): | A sublethal dose of Cd2+ (0.2mg Cd+2 Kg¯1.b.wt.) was subcutaneously injected to pregnant mice on daily basis for 10 consecutive days as from the 3rd day of gestation. |
| Group (3): | The animals received Mg+2 as s.c. injection (73mg Mg+2 kg¯1 b.wt. day¯1) starting from the first day of gestation up to 18 days, this dose proved (according to preliminary experiments) to give the best protective effect against Cd+2 intoxication, when administered for 18 consecutive days starting from the 1st day of gestation |
| Group (4): | The animals received vit. E as repeated s.c. daily doses of 100mg kg¯1 b.wt., in vegetable oil (Shaikh et al., 1999), for the same period of Mg+2 administration. Groups three and four served as negative controls . |
| Group (5): | The animals received the previously mentioned doses of both Cd+2 and Mg+2 with the same routes and manners. |
| Group (6): | The animals received the previously mentioned doses of Cd+2 and vit .E with the same routes and manners. |
Pregnant mice were allowed to deliver normally and lactate their newborns. The litter size (no. of newborns litter-1, as well as the number of dead newborns litter-1 were recorded just after parturition. Morphological examination of the newborns, were performed as described by Wilson 1964 in an orderly manner from head to tail with the help of a magnifying lens at the day of birth. Abnormalities were indicated externally by attenuated abdominal musculature, cleft lip, eye, ear and limb deformities, the number of external malformed newborns litter-1 were recorded. Body weights, linear measurements of the body, hind limb and ear pinna in addition to the number of mortalities were recorded for each individual at the day of birth as well as after two and four postnatal weeks.
Ten newborns were selected from each of the studied groups at the day of birth and prepared for Cd+2 determination. Cadmium level in the whole body of each newborn was estimated by atomic absorption spectrophotometer equipped with an HGA-400 graphite furnace (Prkin-Elmer–Germany).
Ten female mice at the day of parturition as well as ten of the offspring, after two and four postnatal weeks, from each group were anaesthetized, blood samples were withdrawn through a heart puncture and collected in heparinized tubes for hematological examinations. Then centrifuged at 3000 r.p.m. for 15 minutes, the plasma was separated and used for biochemical analysis. Blood was analyzed for the RBCs Hb, hematocrit values (Hct) and leucocytes count (WBCs) using a hematology coulter counter, (Model S 770, coulter Electronics. Inc., Hialeah, FL). Calculations of erythrocytes indices including the determination MCV, MCH and MCHC were performed according to Wintrobe et al. (1981).
Blood smears were stained with Giemsa stain, and the percentage of granulocytes, monocytes and lymphocytes were calculated after counting 200-400 cells (Miale, 1972). Determinations of aminotransferases (AST and ALT) ALP, activities in plasma were performed according to White et al. (1970) and Bauer (1982), respectively. Urea and creatinine concentrations in the plasma were determined using kits purchased from Stabino Chemicals (U.S.A) through Gamma Trade Company (Cairo).
Data were expressed as mean ± S. E. The significance of differences was determined using Student’s “t” test, and level of significance was reported at P < 0.05 (Snedecor, 1971).
Table 1 indicates that Cd+2 injection to pregnant mice significantly increased the number of dead newborns litter-1, externally malformed newborns litter-1 and caused marked decreases in litter size as compared to control group. Also, Cd+2 accumulated in the bodies of newborns as a result of their maternal injection with it.
The data indicated that injection of Mg+2 to animals completely prevented Cd+2-induced defects. While the injection of vit. E. for 18 successive days starting from the 1st day of gestation provided partial protective effect against these defects, since there were a significant differences between the values of this group (Cd+2 + vit .E group), both the control and Cd+2 - treated group. Meanwhile, Cd+2 levels was still high in the bodies of newborns of both (Cd+2 + Mg+2) and (Cd+2 + vit .E.) treated groups .
Newborns maternally treated with Cd+2 show significant decreases in both their body weights and linear lengths, while, the mortality percentages were increased at the day of birth as well as during the first two postnatal weeks and were fixed during the next two postnatal weeks i.e. from the second to the fourth postnatal weeks (Table 2).
In respect to the same Table, Mg+2 administration prevented, completely the offspring growth retardation and mortality. While, vit. E injection showed only a moderate improvement on growth retardation and partially reduced the increases in the percentages of mortality that were induced by maternal Cd+2 administration. Tables 3 and 4 demonstrate that Cd+2 injection elicited marked depression in the number of RBCs, Hb and PCV of female mice at the day of parturition as well as in their offspring after two and four postnatal weeks. While MCV, MCH and MCHC did not differ significantly from control. Co-administration of Mg+2 with Cd+2 succeeded completely in preventing the declines in the mentioned erythrocytic parameters that were induced by the later. On the other hand, vit. E prevented these depressions partially. Tables 5 and 6 indicate that the injection of Cd+2 induced significant increases in the number WBCs and granulocytes, while evoked a marked drop in the percentage of lymphocytes. However, monocyte’s percentage showed insignificant changes in female mice at the day of parturition and their offspring after two and four postnatal weeks. Magnesium administration provided complete protection against Cd+2 induced perturbations in the total numbers of WBCs and their differential counts, while vit. E injection partially prevented these alterations. Regarding Tables (7 and 8) Cd+2 injection caused significant increases in the activities of AST, ALT and ALP in plasma. It also induced marked elevations in urea and creatinine levels in the plasma of females, at the day of parturition, as well as their offspring after two and four postnatal weeks. Magnesium administration succeeded in preventing the increases in the above mentioned biochemical parameters that were induced by Cd+2, while vit. E injection partially improved their levels in the plasma. Animal groups receiving either Mg+2 or vit. E alone did not show any difference in all the studied parameters, with respect to those of the control group.
The data indicate that Cd+2 administration for 10 successive days from the 3rd day of gestation) caused significant toxic effects in pregnant mice and their newborns. It induced significant reduction in the number of fetuses born to treated mothers as indicated by litter size (Table 1). This may be due to the toxic effect of Cd+2 on implantation and/or resorption of implanted embryos. Chiquoine (1965) mentioned that the sensitivity of Cd+2 begins around day 5, the time of implantation in the mouse. Chernoff (1973) observed increased embryonic resorption as a result of subcutaneous Cd+2 administration to pregnant rats. Data showed that Cd+2 injection increased the percentage of fetal as well as postnatal mortalities till the end of the 4th postnatal week (Tables 1,2).
These results are in agreement with finding of Chernoff (1973). He also demonstrated dose-related fetal mortality response as a result of subcutaneous injection of Cd+2 to pregnant rats. The observed increase in fetal death can be attributed to the permeability of the placental barrier to free Cd+2. Since these results showed that Cd+2 was able to cross the placenta and accumulated in the fetus (Table 1). Also, human placental and fetal tissues have been shown to accumulate various metals including Cd+2 (Thuerauf et al., 1975).
In the investigation Cd+2 induced external fetal malformations including, attenuated abdominal musculature, facial and limb malformation. Similar results were recently recorded by Warren et al. (2000). The increases in the percentages of malformations in fetuses maternally treated with Cd+2 can be attributed damage to specific organ systems at critical or most sensitive stage of development at time of Cd+2 insult. The observed malformations may be also, attributed to genotoxicity of Cd+2, since its administration may cause shift mutations (Biggart and Murphy, 1988) and chromosomal aberrations (Hartwig, 1994). It has direct effects on bone tissues. A concentration of 50 nM caused an increase in bone resorption in fetal rat limb cultures (Bhattacharyya et al., 1988). 2.5 uM Cd+2 leads to a decrease in both calcified and uncalcified osteoid tissue in embryonic chick femur cultures (Kaji et al., 1988).
The fetal growth retardates in fetuses maternally treated with Cd+2 (as indicated by decreases in the body weights and linear lengths of the body, hind limb and ear pinna) were detected (Table 2). Lynn et al. (1997) showed that DNA ligation seems to be susceptible to Cd+2 inhibition and that may bind to protein thiols to inhibit DNA repair.
| Table 1: | Means of litter size, dead newborns litter¯1, malformed newborns litter-1 and Cd2+ concentrations in the whole body of newborns in different groups at the day of birth. |
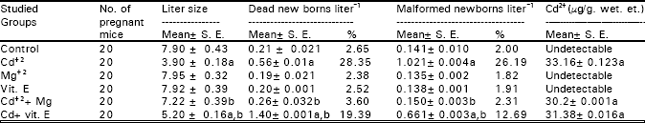 | |
| a, b = Statistically significant (P< 0.05) when compared with values of the control or cadmium injected group, respectively | |
| Table 2: | Means of body weights, lengths of body, hind limb, ear pinna and moralities of the offspring in different groups at birth as well as after two and four postnatal weeks |
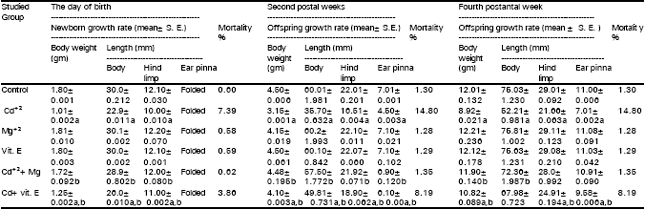 | |
| a, b= Statistically significant (P<0.05) when compared with values of the control of cadmium injected group, respectively | |
| Table 3: | The erythrocytic indices in the blood of female albino mice of different groups at the day of parturition |
 | |
| Data are means of 10 mice ± S. E. a, b= Statistically significant (P<0.05) when compared with values of the control or cadmium injected group, respectively | |
| Table 4: | The erythocytic indices in the blood of the offspring of different groups after two and four postnatal weeks |
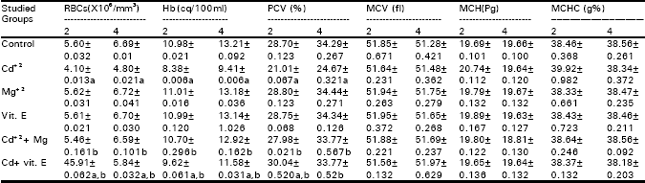 | |
| Data are means of 10 mice ± S.E. a,b = Statistically significant (P<0.05) when compared with value sof the control of cadmium injected group, respectively | |
| Table 5: | The Total and differential leucocytic counts in the blood of female albino mice in different group at the day of parturition |
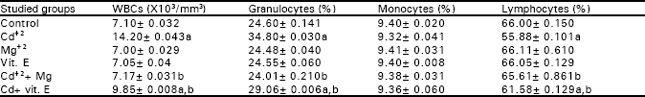 | |
| Data are means of 10 mice ± S. E. a,b = Statistically significant (P<0.05) when compared with values of the control or cadmium injected group, respectively | |
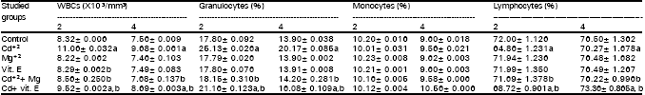
| Table 6: | The total and differential leucocytic counts in the blood of the offspring in different groups after two and four postantal weeks. |
 | |
| Data are means of 10 mice ± S. E. a,b = Statistically significant (P<0.05) when compared with values of the control or cadmium injected group, respectively | |
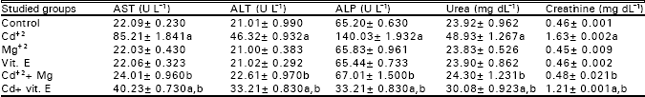
| Table 7: | Levels of some biochemical parameters in the plasma of female albino mice in different groups at the day of parturition |
 | |
| Data are means of 10 mice ± S. E. a,b = Statistically significant (P<0.05) when compared with values of the control or cadmium injected group, respectively | |
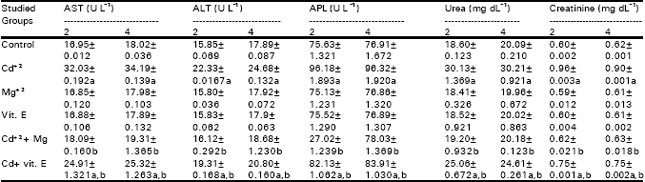
| Table 8: | Levels of some biochemical parameters in the plasma of the offspring in different groups after two and four postnatal weeks |
 | |
| Data are means of 10 mice ± S.E. a,b = Statistically significant (P<0.05) when compared with value sof the control of cadmium injected group, respectively | |
Thus, fetal growth retardation may result from either the cadmium-induced disturbance of the maternal-embryonic relationship and interference with fetal nutrition or a direct effect of Cd+2 on fetal tissues.
Normocytic normochromic anaemia was detected in female mice of Cd+2 treated group at the day of parturition and their offspring after two and four postnatal weeks as evidenced by decreases in RBCs counts, Hb and PCV and along with insignificant changes in MCV, MCH and MCHC (Tables 3 and 4). These results confirmed those reported by El-Sebai et al. (1994) on adults. As regard to the offspring, Smith et al. (1991) fed Holstein heifers a mixed diets containing 0.25, 1.0 and 5.0mg Cd+2 kg-1 of the diet mixture (control, low – and high – Cd), respectively for 394 days before parturition. They observed that calves from dams fed on high -Cd+2 diet showed a reduction in Hb content amounting to 18%.
Anaemia is caused by
| (a) | blood loss, which may be acute or chronic, |
| (b) | defective red cell production due to lack of essential nutrients or congenital or acquired disorders of erythropoiesis, and |
| © | impaired red cell survival due to hemolysis (Campbell et al., 1984). |
Thus, the depression in RBCs count recorded in the present work could be due to disturbed hematopoieses, destruction of cells and/or their enhanced removal from circulation.
Leucocytosis, granulocytosis and lymphopenia were observed in female mice as well as in their offspring, as a result of Cd+2 administration while, monocytes did not show significant alterations as compared to control (Tables 5 and 6). El-Sebai et al. (1994) observed that Cd+2 administration caused an increase in WBCs in broilers as a result of feeding them a basal diets containing 0, 50, 75 and 100mg Cd+2kg¯1 as CdCl2 untill 6 weeks old. Kataranovski et al. (1998) detected an increase in the number of granulocytes following Cd+2 administration in rats. Liu et al. (1999) demonstrated marked increases in the blood neutrophils (>7.5-fold) of mice that were exposed to Cd+2 via s.c. injection, with a wide range of doses.
The number of granulocytes in the blood is usually maintained with close limits. Mature cells are released at a steady rate under normal circumstances. Certain infections can result in the release from bone marrow of more immature cells resulting in a rise in peripheral white count. The average lifespan of the mature granulocyte is only 12 hours (Campbell et al., 1984). Cadmium is also reported to stimulate cytokine release from cells in other tissues, including lung alveolar macrophages (Driscoll et al., 1992) and neutrophils (Horiguchi et al., 1993) Lymphopenia occurs with almost any form of stress, it accompanies most acute infections and it is a feature of some of the immune deficiency states. Thus the observed depression in lymphocytes count may reflect disturbance and stress on the immune system in response to Cd+2 intoxication. From the data, it is obvious that Cd+2 administration induced significant increases in the activities of AST and ALT in the plasma of female mice and their offspring as compared to their relative control (Tables 7 and 8).
These results are in agreement with those recorded on adult experimental animals. Dudley et al. (1982) reported an increased plasma levels of AST and ALT associated with Cd intoxication in rats. Also, Groten et al. (1994) noticed increased plasma transaminases in male Wistar rats fed on diets containing Cd+2 30mg kg¯1 as CdCl2 for 8 weeks. In addition, Novelli et al. (1998) reported increased ALT activity in rats that were exposed to CdCl2 for 7 days, in drinking water (100 mg l¯1).
Increased serum transaminase activity in liver disease is assumed to reflect leaking from injured cells (Wyngaarden and Smith, 1985). Sauer et al. (1997) mentioned that rats administrated CdCl2 showed clear signs of liver injury, as measured by plasma ALT activity and liver pathology. Thus, the elevations in transaminases activities, detected is due to Cd+2-induced hepatotoxicity.
Cd+2 injection evoked marked increases in the activities of ALP in the plasma of female mice and their offspring as compared to controls (Tables 7 and 8). Similar increases in plasma ALP activities as a result of Cd+2 administration to adult animals were recorded in previous (Claverie et al., 2000). ALP found in many tissues, including liver, bile ducts, intestine, bone, kidneys, placenta and leucocytes. An increase, it is found in hepatocellur diseases and cholestasis (Kaneko, 1989). The increased levels of ALP that detected in the plasma of female mice and their offspring may be due to hepatocellur damage in response to Cd+2 injection. Kamiyama et al. (1995) reported that Küpffer cells are stimulated to produce cytokines such as TNF-α and 1L-6 after cd administration, and these cytokines are responsible for certain manifestations of liver damage by cadmium.
In the investigation, significant increases in plasma urea and creatinine levels were observed in females treated with Cd+2 and their offspring (Tables 7 and 8). In agreement with these resutls, Smith et al. (1991) observed increases in blood urea nitrogen by 63% in calves from Holstein heifers dams given high cadmium diets (5.0mg Cd kg¯1) for long period before parturition.
Plasma urea acts as a rough guide of renal function. A decrease in the rate of excretion of urea produces an increase in the concentration of plasma urea (Kaneko, 1989). The measurement of plasma creatinine which is closely related to glomerular filteration rate, is preferable as an indicator of renal function (Campbell et al., 1984). The observed significant elevations in the levels of both plasma urea and creatinine reflect the impairment of renal function.
Cadmium, like most heavy metals, is not a biotransformed, and hence biologic tolerance must, therefore, be of different nature than breakdown into less toxic or more readily excreted products. In all tissues Cd+2 induces and binds to metallothionine (MT), a low-molecular-weight scavenger protein, and is stored as a nontoxic CdMT complex (Webb and Cain, 1986). Over time, CdMT is translocated from liver to the kidneys due to normal turnover of hepatocytes as well as hepatic injury (Chan et al., 1993). CdMT is filtered from the blood and efficiently reabsorbed by the renal proximal tubules (Foulkes, 1978). The reabsorbed CdMT is quickly degraded by lysosomal enzymes, liberating Cd ions that bind to preexisting or newly synthesized renal MT (Squibb et al., 1979). Toxicity may result when MT synthesis becomes insufficient to keeps up with the demand and the non-MT bound Cd eventually over helms other defense mechanisms as well and causes toxicity (Goyer et al., 1989).
Cadmium exposure also affects tissue glutathione (GSH) levels, which is the major cellular thiol conductant that is involved in metal intoxication by forming GSH metal-1 ion complex. Singhal et al. (1987) reported that depletion of that GSH enhanced the toxicity of Cd+2 and elevation of tissue GSH levels protected against its toxicity. Rana and Verma (1996) mentioned GSH decreased the renal Cd accumulation in rats chronically exposed to it.
In this investigation, Mg+2 administration succeeded in projecting female mice and their offspring against Cd+2-induced toxicities, since, it completely prevented embryo, haemato, hepato and nephrotoxicities, while the concentration of Cd+2 was still high levels in the bodies of the newborns. As an essential metal for cell metabolism, Mg+2 antagonism has been attributed to its competition with other heavy metals for high affinity binding sites on DNA, proteins, or phosphorylated compounds (Littlefield et al., 1994). Li et al. (1996) observed that low doses of Mg+2 slightly increase cellular GSH. They concluded that Mg+2 may stimulate de novo GSH biothynthesis, since many enzymes require Mg+2 as a cofactor for their activities.
Thus, the detected protective action of Mg+2 against the previously mentioned toxic effects of Cd+2 may be due to either the antagonistic effect of Mg+2 and/or the stimulatory effect of Mg+2 in producing de novo GSH.
Vit. E administration improved the Cd+2-induced embryo toxicity as well as haemato, hepato and renal toxicities partially, along with high levels of Cd+2 in the bodies of newborns. Selenke and Foulkes (1981) mentioned that CdMT binds to the brush border membrane of proximal tubular epithelial cells and is reabsorbed via endocytosis, lipid bilayer of the brush border membrane may be disturbed during CdMT uptake. Also, Cd+2 interferes with mitochondrial function and causes lipid peroxidation (Koizumi et al., 1994), resulting in oxidative stress to the cell.
Vitamin E exists mainly in cell membranes and serves as a radical scavenger protecting the cell membrane against the propagation of lipid peroxidation. The membrane lipid-stabilizing effect of vit. E can occur by stabilizing the lipid bilayer through a Van der Waals interaction of tocopherol with unsaturated fatty acids of phospholipid or by stabilizing to polypeptide chains of membrane proteins (Buttress and Diplock, 1986). Tandon et al. (1992) mentioned that vit. E is not likely to directly bind to Cd ions and is therefore, its protective effect is most likely due to its antioxidant property. Vit. E provided a marked reduction rather complete suppression of Cd+2-induced toxicities. This lack of effect of vit. E may be attributed to the presence of other mechanisms of Cd+2-toxicity that are not influenced by the antioxidant.
Cadmium concentrations were still at high levels in the bodies of the newborns of (Cd+2 + Mg+2) and (Cd+2+ vit. E)- treated groups. Thus, the complete as well as the partial protection against Cd+2-induced embryo toxicity offered by Mg+2 and vit. E, respectively, is achieved in some manner other than effecting a biochemical Cd+2 block in the placenta, or in altering the permeability of the placenta.
It is to be concluded that Mg+2 is more effective than vit. E in protecting female mice and their offspring from Cd+2-induced intoxication. Mg+2 proved its ability as a protective agent against Cd+2-induced toxicities.