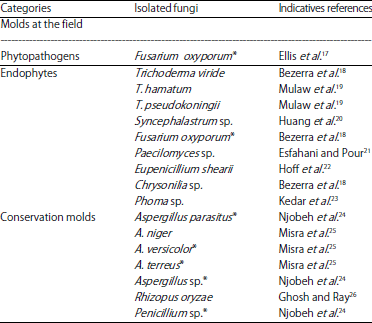
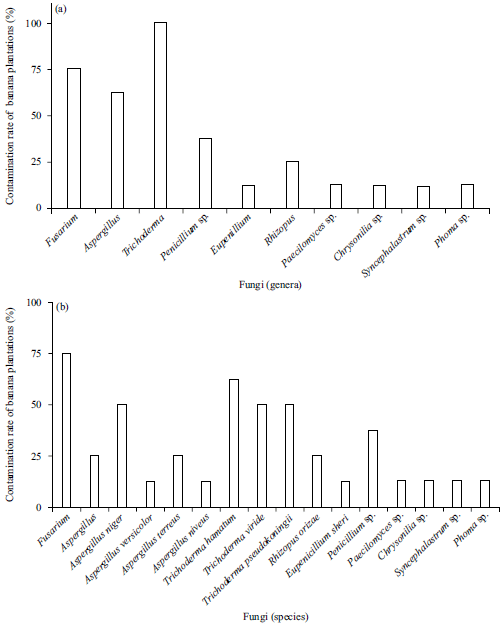
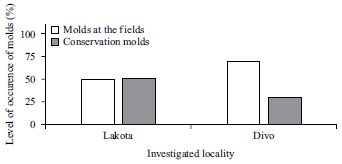
Research Article
Cropping Practices and Fungal Contamination in Banana Plantations in Côte d’Ivoire
Food, Bioprocess, Toxicology, Environments (Team 4651)-Food Matrix and Microbiota-Campus 1, Institute for Fundamental and Applied Biology, University of Caen Normandie, Esplanade de la Paix, 14032 Caen cedex 5, France
LiveDNA: 225.16450
Jean-Michel Panoff
Food, Bioprocess, Toxicology, Environments (Team 4651)-Food Matrix and Microbiota-Campus 1, Institute for Fundamental and Applied Biology, University of Caen Normandie, Esplanade de la Paix, 14032 Caen cedex 5, France
Stephanie Gente
University of Caen Normandie, Food, Bioprocess, Toxicology, Environments (Team 4651)- Environmental Toxicology, Aerial Environments and Cancer, 14000 Caen, France
David Garon
University of Caen Normandie, Food, Bioprocess, Toxicology, Environments (Team 4651)- Environmental Toxicology, Aerial Environments and Cancer, 14000 Caen, France
Jean-Philippe Rioult
University of Caen Normandie, Food, Bioprocess, Toxicology, Environments (Team 4651)- Environmental Toxicology, Aerial Environments and Cancer, 14000 Caen, France
Tia Jean Gonnety
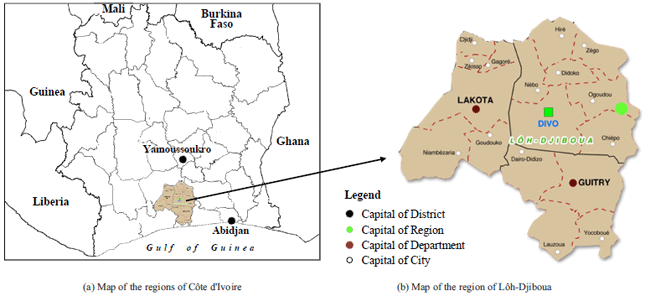
Laboratory of Biocatalysis and Bioprocessing, Faculty of Science and Food Technology, Nangui Abrogoua University, 02 BP 801 Abidjan 02, Côte d'Ivoire
Marina Koussémon
Laboratory of Biotechnology and Food Microbiology, Faculty of Science and Food Technology, Nangui Abrogoua University, 02 BP 801 Abidjan 02, Côte d’Ivoire