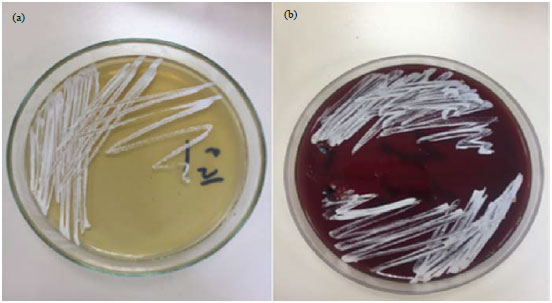
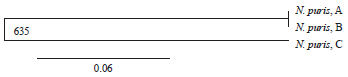
Case Study
First Documented Mastitis Case of Nocardia puris in Turkey from Two Cows: Microbiological and Molecular Identification
Department of Microbiology, Faculty of Veterinary Medicine, Mehmet Akif Ersoy University, 15030, Burdur, Turkey
LiveDNA: 90.25177
Dilek Ozturk
Department of Microbiology, Faculty of Veterinary Medicine, Mehmet Akif Ersoy University, 15030, Burdur, Turkey
Mehmet Kaya
Department of Microbiology, Faculty of Veterinary Medicine, Mehmet Akif Ersoy University, 15030, Burdur, Turkey