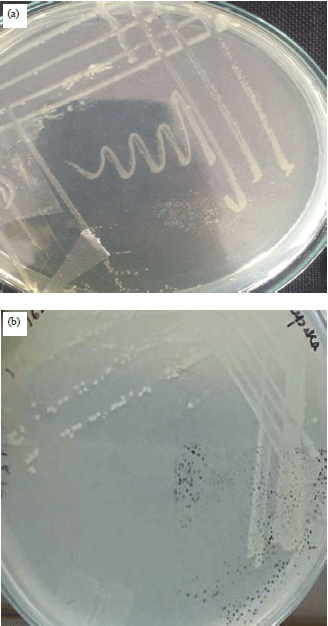
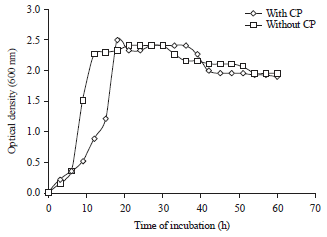
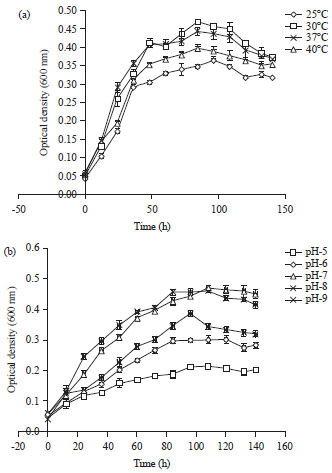
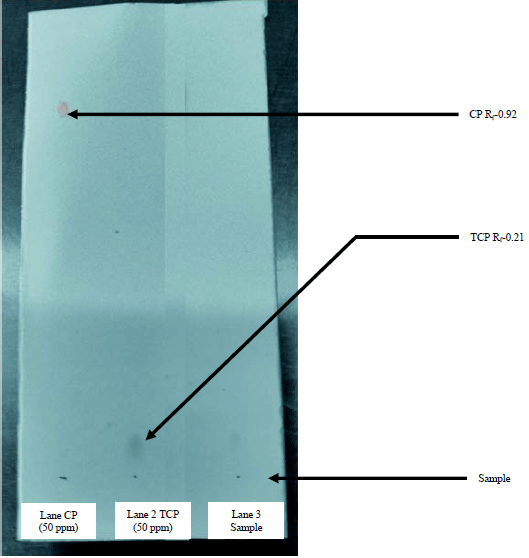
Research Article
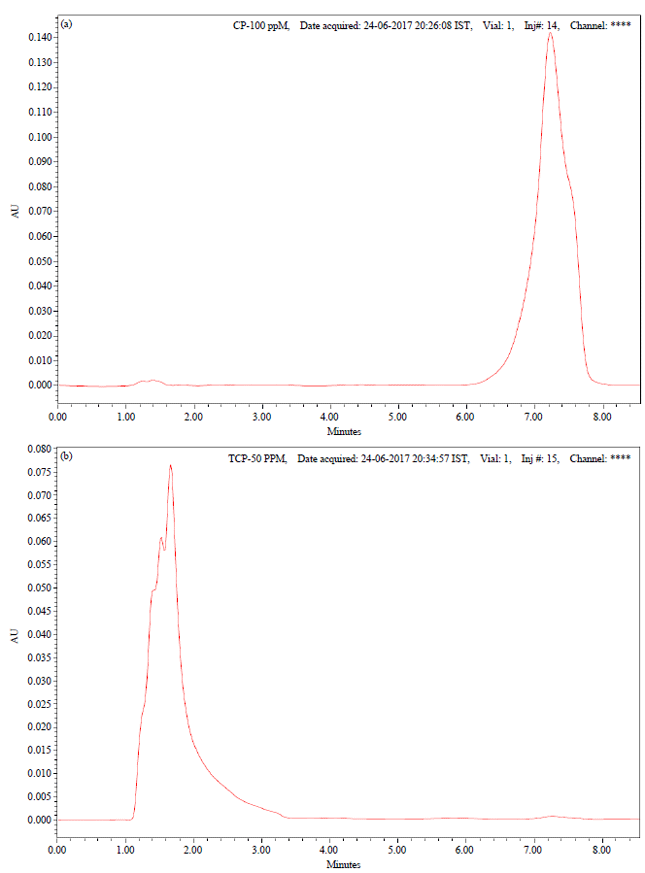
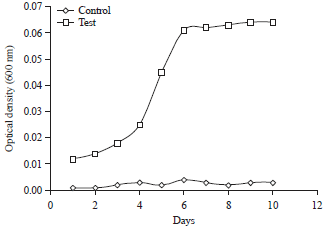
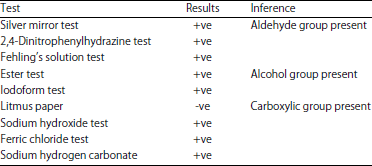
Proficient Biodegradation Studies of Chlorpyrifos and its Metabolite 3,5,6-Trichloro-2-pyridinol by Bacillus subtilis NJ11 Strain
Laboratory of Biosensor Technology, Department of Biotechnology, Punjabi University, 147002 Patiala, Punjab, India
LiveDNA: 91.1587
Neelam Verma
Laboratory of Biosensor Technology, Department of Biotechnology, Punjabi University, 147002 Patiala, Punjab, India
LiveDNA: 91.19908