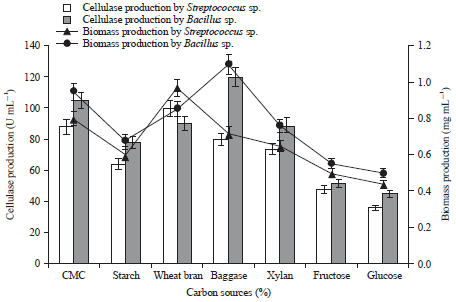
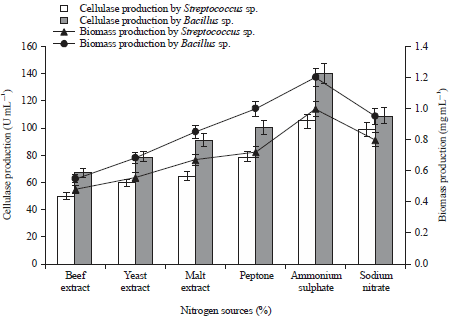
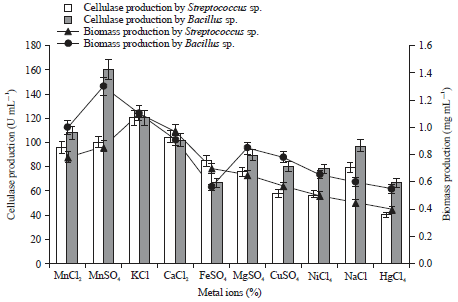
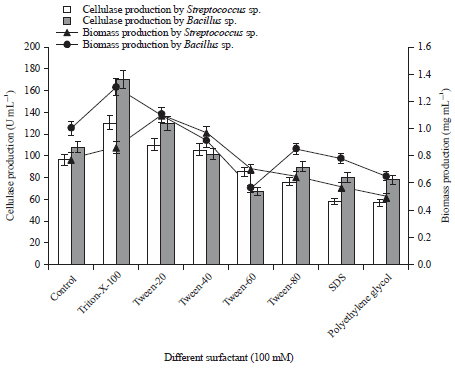
Research Article
Sugarcane Baggase Agro-waste Material Used for Renewable Cellulase Production from Streptococcus and Bacillus sp.
Department of Microbiology (Centre of Excellence), Dr. Ram Manohar Lohia Avadh University, 224001 Faizabad, Uttar Pradesh, India
Pooja Pathak
Department of Microbiology (Centre of Excellence), Dr. Ram Manohar Lohia Avadh University, 224001 Faizabad, Uttar Pradesh, India
Rajeeva Gaur
Department of Microbiology (Centre of Excellence), Dr. Ram Manohar Lohia Avadh University, 224001 Faizabad, Uttar Pradesh, India
LiveDNA: 91.16855