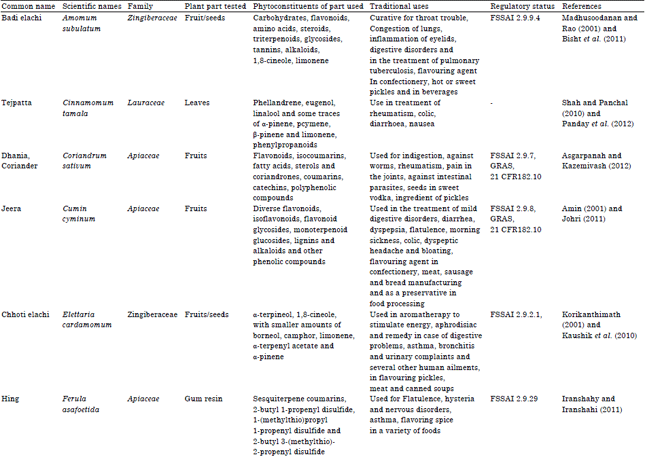
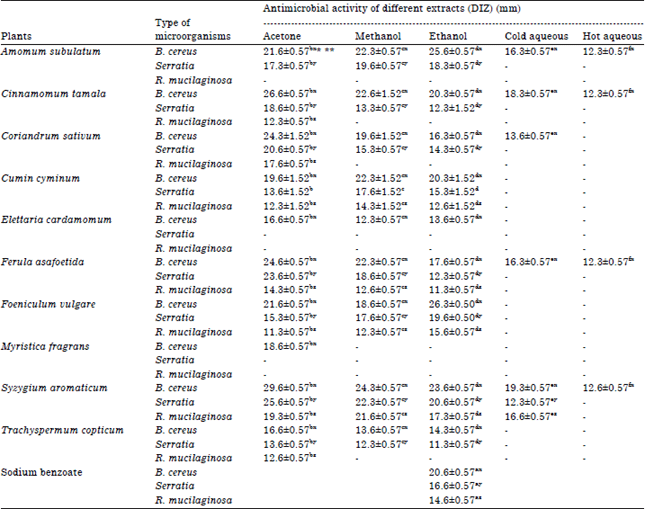
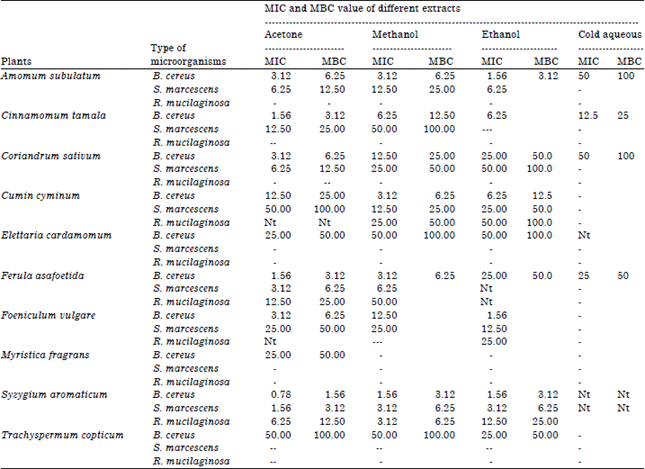
Research Article
Comparative Evaluation of Antimicrobial Activities of Commonly Used Indian Spices Against Microbes Associated with Juices
Department of Microbiology, Kurukshetra University, Kurukshetra, 136119, India
Neeraj Kumar Aggarwal
Department of Microbiology, Kurukshetra University, Kurukshetra, 136119, India
Manpreet Kaur
Department of Microbiology, Kurukshetra University, Kurukshetra, 136119, India