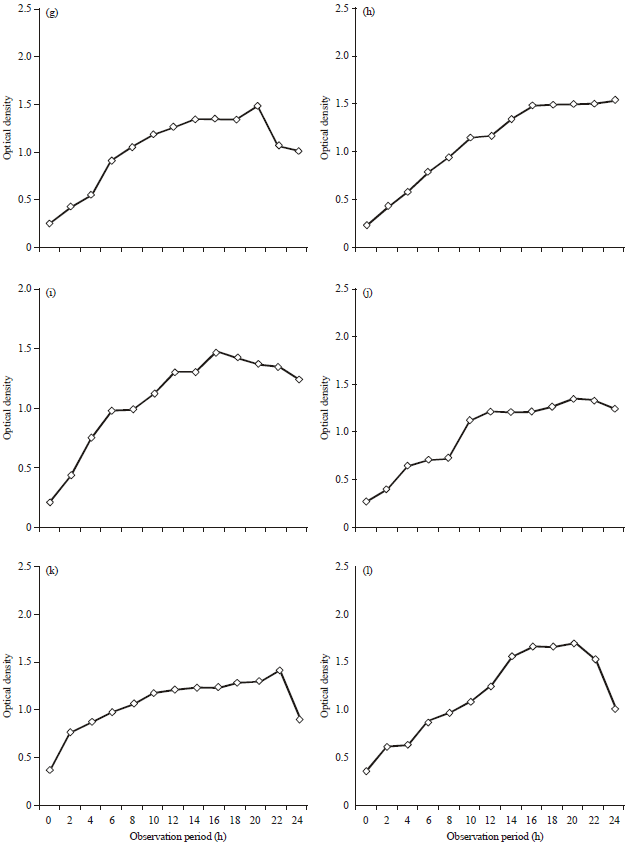
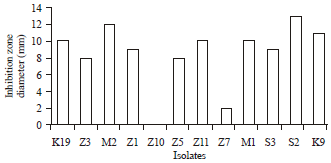
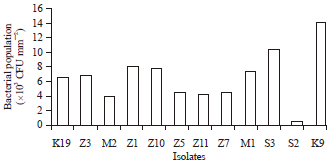
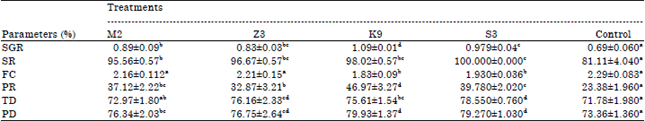
Research Article
Screening of Probiotic Bacteria Candidates from Gastrointestinal Tract of Pacific White Shrimp Litopenaeus vannamei and their Effects on the Growth Performances
Department of Aquaculture, Faculty of Fisheries and Marine Science, Bogor Agricultural University, Jalan Agatis, Dramaga Campus, Bogor, West Java, 16680, Indonesia
Tita Nopitawati
Study Program of Aquaculture, Graduate School, Bogor Agricultural University, Indonesia
Dedi Jusadi
Department of Aquaculture, Faculty of Fisheries and Marine Science, Bogor Agricultural University, Jalan Agatis, Dramaga Campus, Bogor, West Java, 16680, Indonesia