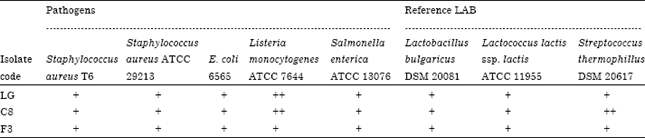
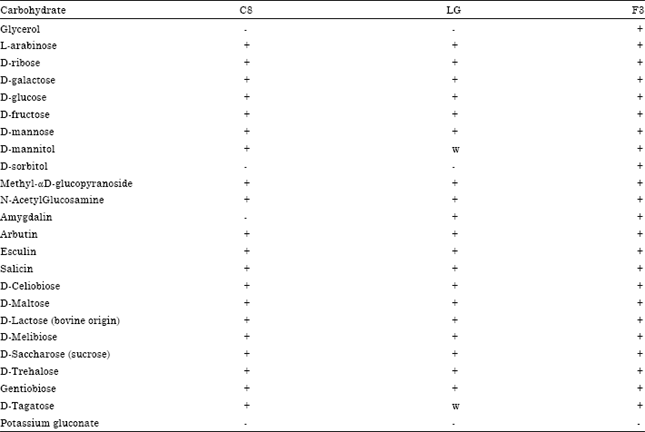
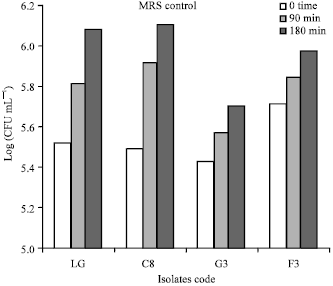
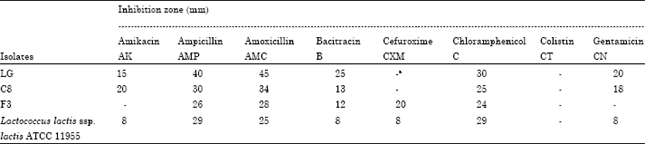
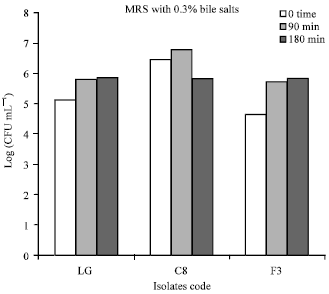
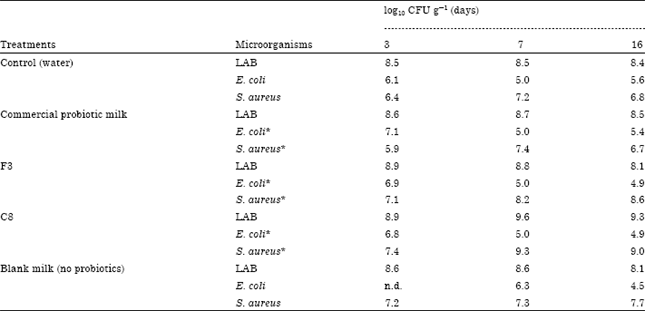
Research Article
Camel's Milk as a Natural Source for Probiotics
Department of Food and Nutrition Sciences, College of Agricultural and Food Sciences, King Faisal University, P.O. Box 400, Al-Ahsa, Saudi Arabia
Najeeb S. Al-Zoreky
Department of Food and Nutrition Sciences, College of Agricultural and Food Sciences, King Faisal University, P.O. Box 400, Al-Ahsa, Saudi Arabia
Hassan A. El-Dermerdash
Department of Food and Nutrition Sciences, College of Agricultural and Food Sciences, King Faisal University, P.O. Box 400, Al-Ahsa, Saudi Arabia