Research Article
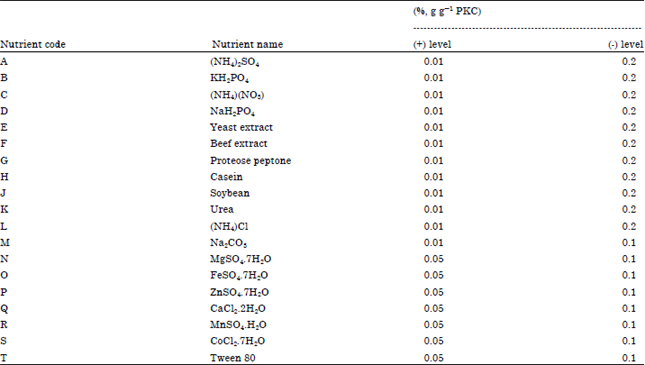
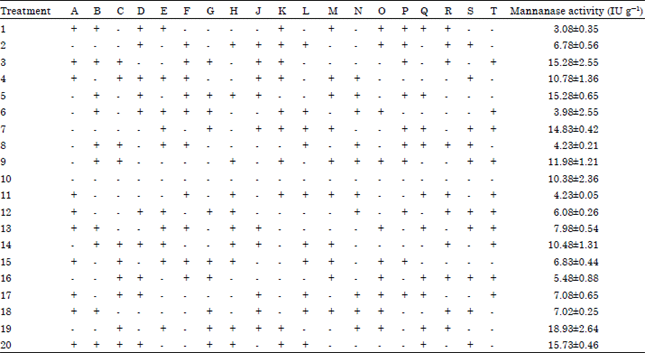
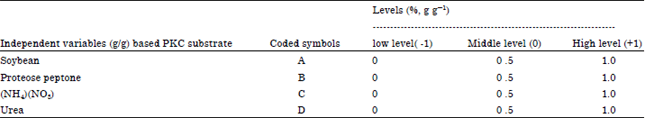
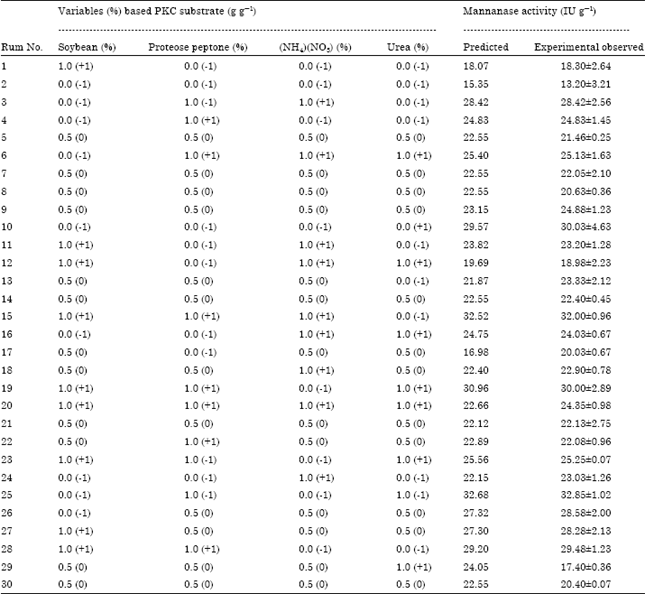
Screening and Optimization of Medium Composition for Mannanase Production by Aspergillus terreus SUK-1 in Solid State Fermentation Using Statistical Experimental Methods
Department of Chemistry and Biology, Centre for Defense Foundation Studies, National Defense University of Malaysia, Sungai Besi Camp, 57000, Kuala Lumpur, Malaysia
Noraini Samat
Strategic Livestock Research Centre, Malaysian Agriculture Research and Development Institute, P.O. Box 12301, 50774 Kuala Lumpur, Malaysia
Wan Mohtar Wan Yusoff
School of Bioscience and Biotechnology, Faculty of Science and Technology, National University of Malaysia, 43600, Bangi, Malaysia