Research Article
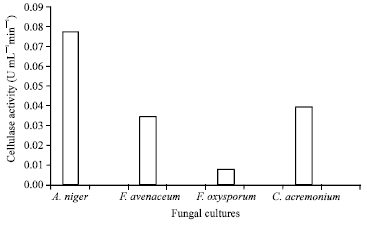
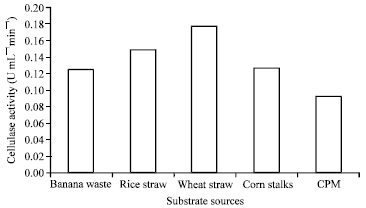
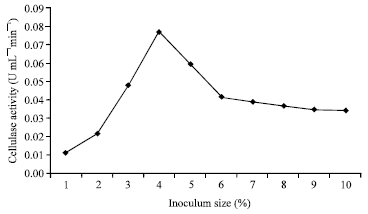
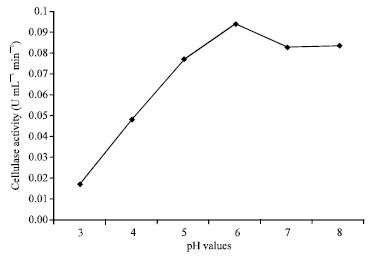
Optimization of Culture Conditions Affecting Fungal Cellulase Production
Department of Dairy Science, National Research Center, Dokki, Giza, Egypt
H.A. Murad
Department of Dairy Science, National Research Center, Dokki, Giza, Egypt
A.M. Kholif
Department of Dairy Science, National Research Center, Dokki, Giza, Egypt
M.A. Hanfy
Department of Animal Production, Faculty of Agriculture, Cairo University, Giza, Egypt
M.H. Abdel Gawad
Department of Animal Production, Faculty of Agriculture, Cairo University, Giza, Egypt