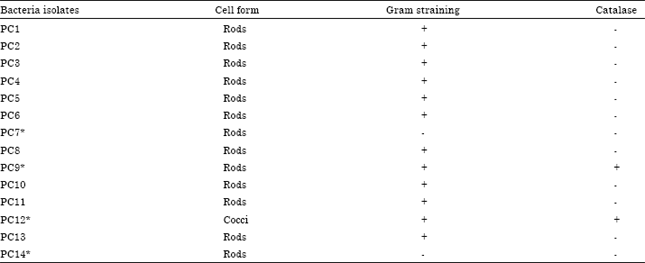
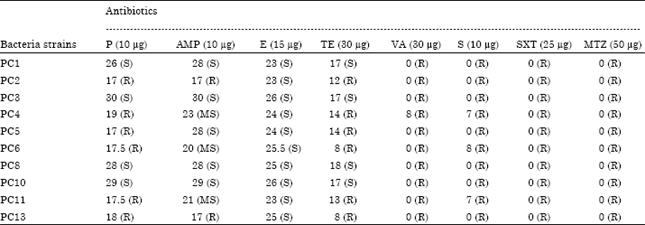
Research Article
Antibiotic Resistance of Lactic Acid Bacteria Isolated from a Fermented Fish Product, Pla-chom
Department of Veterinary Public Health, Faculty of Veterinary Medicine, Khon Kaen University, Khon Kaen, 40002, Thailand
V. Leelavatcharamas
Fermentation Research Center for Value Added Agricultural Products, Faculty of Technology, Khon Kaen University, Khon Kaen, 40002, Thailand
P. Sukon
Department of Anatomy, Faculty of Veterinary Medicine, Khon Kaen University, Khon Kaen, 40002, Thailand
S. Yowarach
Department of Veterinary Public Health, Faculty of Veterinary Medicine, Khon Kaen University, Khon Kaen, 40002, Thailand