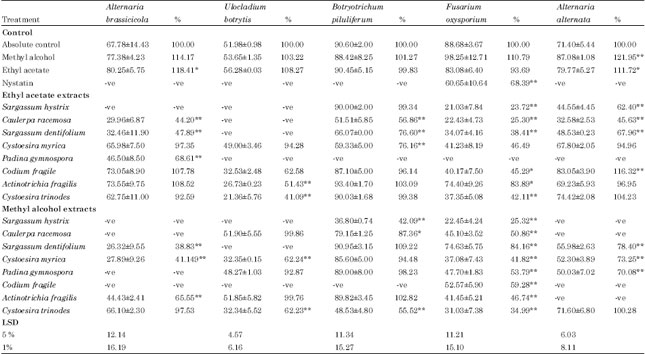
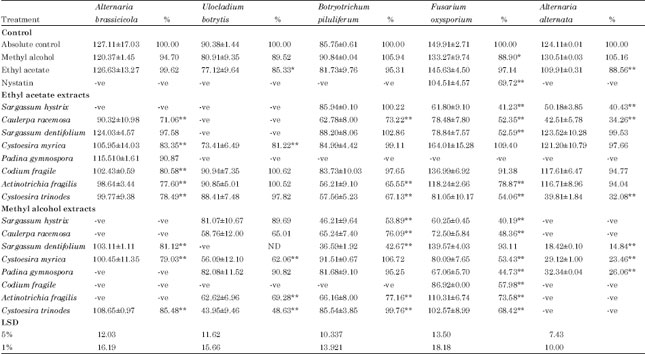
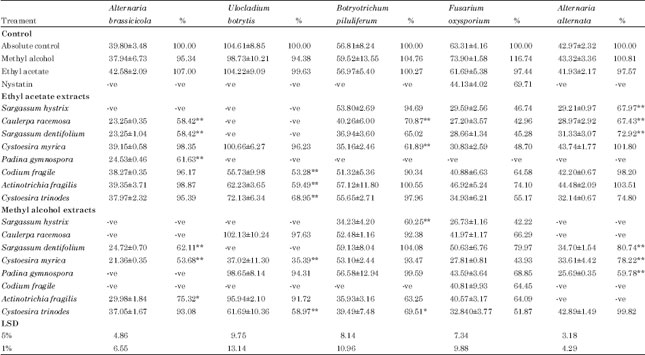
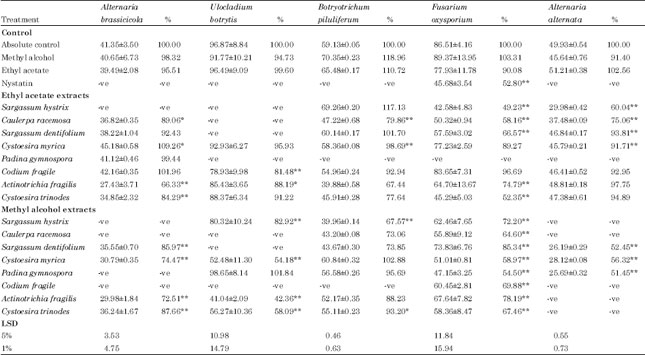
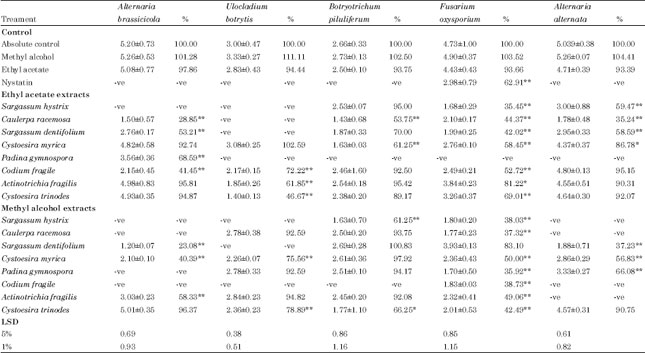
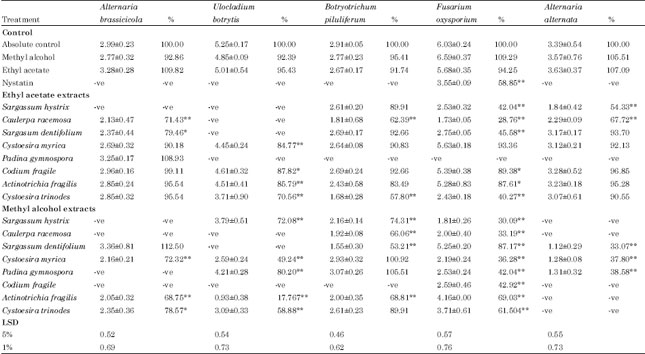
Research Article
Biological Control of Some Pathogenic Fungi using Marine Algae Extracts
Department of Botany, Faculty of Science at Qena, South Valley University, 83523, Egypt
W.M. Salem
Department of Botany, Faculty of Science at Qena, South Valley University, 83523, Egypt
F. Nasr El-Deen
Department of Botany, Faculty of Science at Qena, South Valley University, 83523, Egypt