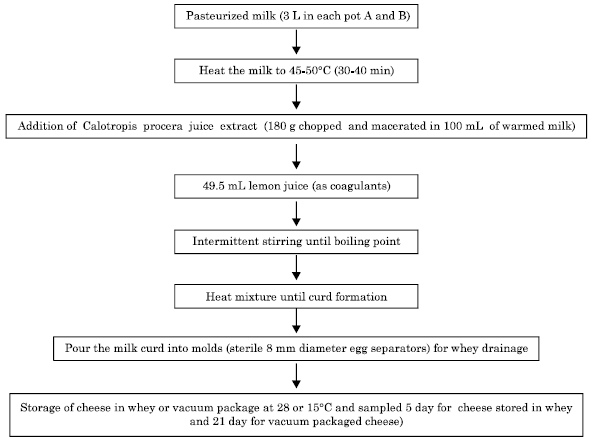
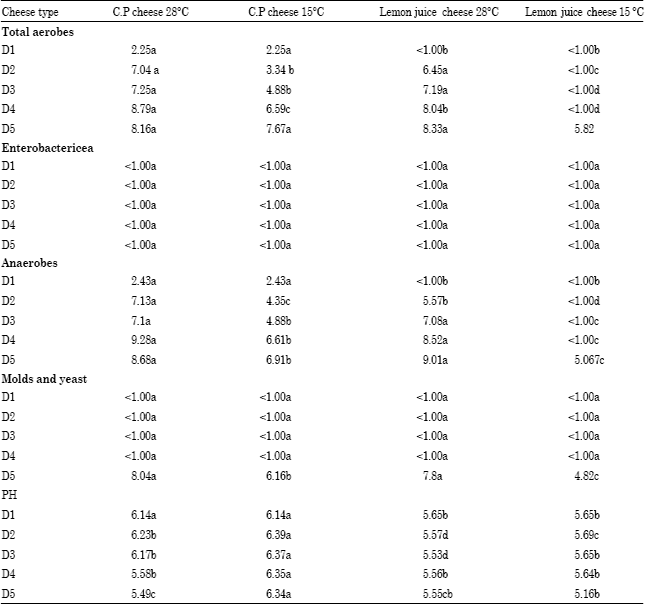
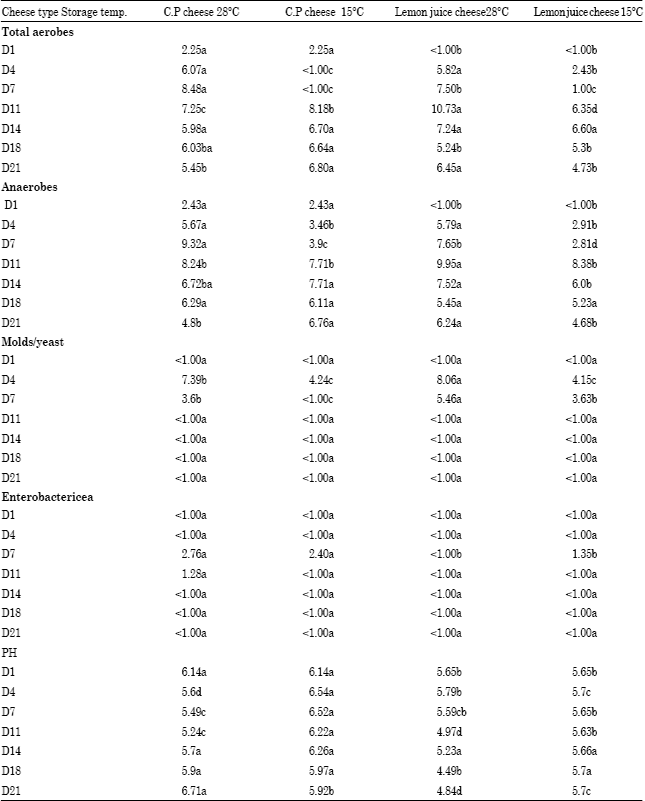
Research Article
Effect of Temperature and Modified Vacuum Packaging on Microbial Quality of Wara A West African Soft Cheese
Department of Veterinary Public Health and Preventive Veterinary Medicine, University of Ibadan, Ibadan, Nigeria
J. Chen
Department of Food Science and Technology, The University of Georgia Griffin GA 30223-1797 USA