Research Article
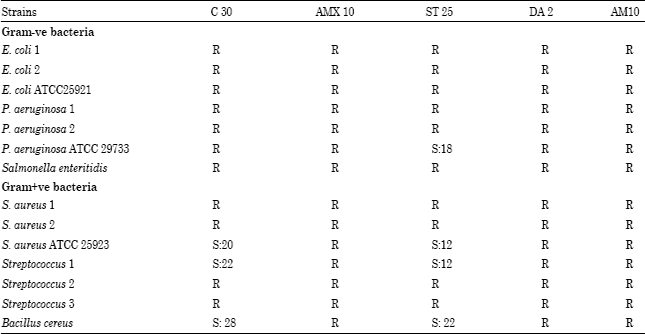
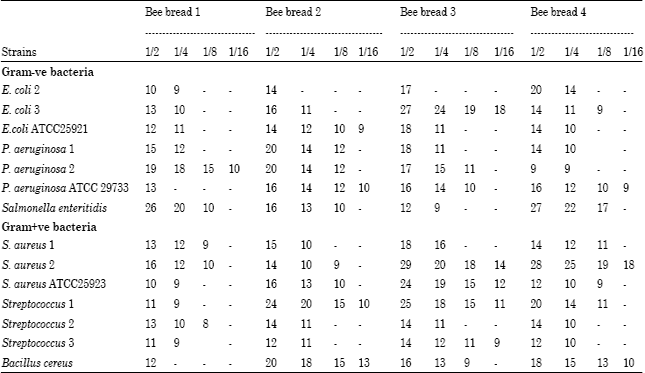
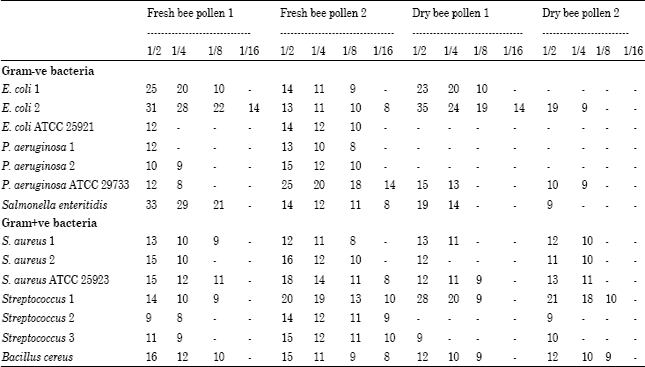
The Antibacterial Activity of Moroccan Bee Bread and Bee-Pollen (Fresh and Dried) against Pathogenic Bacteria
Department of Biology, College of Sciences, Ibn Toufail University, P.O. Box 133, Kenitra, Morocco
I. Zerdani
Department of Biology, Faculty of Sciences Ben Msik, Hassan II-Mohammedia University, P.O. Box 7955, Casablanca, Morocco
I. Kalalou
Department of Biology, College of Sciences, Ibn Toufail University, P.O. Box 133, Kenitra, Morocco
M. Faid
Department of Food Engineering and Technology, Hassan II Institute of Agronomy and Veterinary, P.O. Box 6202, Rabat-Institute, Morocco
M.T. Ahami
Department of Biology, College of Sciences, Ibn Toufail University, P.O. Box 133, Kenitra, Morocco