ABSTRACT
Hexane, chloroform, methanol and water extracts of aerial part of a high altitude Himalayan medicinal plant Valeriana wallichii DC. (Valerianaceae) were tested for their in vitro antimicrobial properties against animal and plant pathogenic bacteria using disc diffusion method. Aqueous extract showed the highest activity against Staphylococcus aureus (23±1.0 mm zone of inhibition, MIC 250 and MBC 500 μg mL-1) followed by methanol extract against Bacillus subtilis (20±1.0 mm ZOI, MIC 31.25 μg mL-1 and MBC 500 μg mL-1), Staphyloccocus aureus (19±0.8 mm ZOI, MIC and MBC 125 μg mL-1) and hexane extract against Bacillus subtilis (18±1.2 mm ZOI, MIC and MBC 125 μg mL-1). Chloroform extract showed the least activity against the test bacteria. The inhibitory potential of V. wallichii extracts was found very notable as compared to Ampicillin (10 mcg) and Erythromycin (15 mcg) which were used as positive control against these tested microorganisms and therefore this plant can be used as a good source of antibiotic substances for possible treatment of bacterial infections of both plants and animals.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=jm.2011.289.296
INTRODUCTION
Healing potential of plant extracts is well known fact and antimicrobial principle is one of the element besides other responsible for the healing. During the last one decade the pace of development of new antimicrobial drugs has slowed down while the prevalence of resistance (especially multiple) has increased tremendously (Hugo and Russell, 1984). Literature reports and ethnobotanical records suggest that plants are the sleeping giants of pharmaceutical industry (Hamburger and Hostettmann, 1991). They may provide novel or lead compounds which may be used as a natural source of antimicrobial drugs in controlling some diseases of plants and animals.
The plant-based, traditional medicine system plays an essential role in health care, with about 80% of the world’s inhabitants relying mainly on traditional medicines for their primary health care (Owolabi et al., 2007). According to World Health Organization (WHO), medicinal plants would be the best source to obtain a variety of drugs. Therefore, such plants should be investigated to better understand their properties, safety and efficacy (Nascimento et al., 2000).
To develop alternative antimicrobial drug, one approach is to screen local medicinal plant, which represent a rich source of novel antimicrobial agents (Khulbe and Sati, 2009). The Himalaya is comprises of number of medicinal plants which are frequently been reported for their traditional uses in the treatment of various ailments (Sati and Joshi, 2010).
Valeriana wallichii DC. (Syn: V. jatamansi Jones) is one of these. It belongs to family Valerianaceae commonly called as Indian valerian. It is indigenous to the temperate Himalayas and found in India, Nepal, Bhutan, Burma, Pakistan and Afghanistan. The plant is widely known for its use in anxiety, insomnia, epilepsy, failing reflexes, hysteria, neurosis and sciatica (Nadkarni, 1976; Baquar, 1989). It is also considered useful as potent tranquilizer, emmenagogue (Nadkarni, 1976), diuretic (Said, 1970) hepatoprotective, diarrhoea (Awan, 1990), gastrospasms (Kapoor, 1990) and hypertension (Chevallier, 1996). However the antibacterial activity of this plant has not been adequately explored. Therefore, in the present investigation antibacterial potential of V. wallichii following standard methodology (disk diffusion method and serial dilution) is explored.
MATERIALS AND METHODS
Plant material and collection: Valeriana wallichii DC. (Valerianaceae) is a small perennial herb of 15-45 cm height, with root stock, thick branching stem, sharply pointed leaves, white or pink flowers in clusters and hairy fruit (Fig. 1). The undergrounds are bitter in taste and contain a characteristic smell. Aerial parts of V. wallichii were collected between July 2006-2007 from their natural sources in different regions of Kumaun Himalaya, India and authenticated by the Department of Botany, Kumaun University, Nainital. The plant was authenticated by Prof. Y.P.S. Pangtey and a voucher specimen was deposited in the herbarium of the Department (KU-101).
Extraction procedure: Aerial part of the plant were thoroughly washed and dried under shade at the room temperature (20±2°C). The dried material was powdered in an electric grinder. To prepare stock solution 50 g of this powder was added to 200 mL of solvents (w/v, 50 g 200 mL-1). Solvents used for extraction were methanol, chloroform, hexane and water. Each extract was shaken for at least 6 h and after that each extract was passed through Whatman filter paper No. 1. The final filtrate as 25% crude extract thus concentrated on a rotary evaporator under vacuum at 20°C was used for the experiments.
Microorganisms used: Eight (Gram +ve and -ve) bacteria (Bacillus subtilis MTCC No. 121, Escherichia coli MTCC No. 40, Agrobacterium tumefaciens MTCC No. 609, Staphylococcus aureus MTCC No. 87, Proteus vulgairs MTCC No. 426, Xanthomonas campestris MTCC No. 2286 borrowed from Institute of Microbial Technology, Chandigarh, India and Xanthomonas phaseoli and Erwinia chrysanthemi were obtained from Plant Pathology Department, G.B. Pant University, Pantnagar, India) were used in this investigation.
 | |
| Fig. 1: | V. wallichii DC |
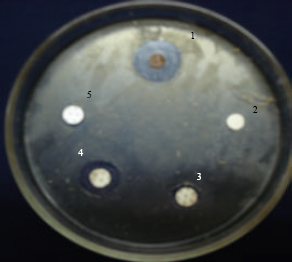 | |
| Fig. 2: | Application of test and control Discs: 1: Extract; 2: Negative control; 3: Erythromycin; 4: Gentamicin and 5: Ampicillin |
Screening of antibacterial activity: Antibacterial tests of selected microorganisms were carried out using disc-diffusion method (Bauer et al., 1966). Nutrient agar plates (90 mm size) were prepared and cooled down at room temperature (20±2°C). A small sterile cotton swab was dipped into the 24 h old culture of bacteria and was inoculated by streaking the swab over the entire agar surface. This process was repeated by streaking the swab 2 or more times rotating the plates approximately 60° each time to ensure even distribution of inoculum. After inoculation the plates were allowed to dry at room temperature (20±2°C) for 15 min in laminar chamber for settle down of inoculum. The filter paper discs (5 mm diam) loaded with 40 μL of extract were placed on the surface of the bacteria seeded agar plates and it was allowed to diffuse for 5 min then these plates were incubated at 37±1°C for 24 h.
Gentamycin (30 mcg), erythromycin (15 mcg) and ampicillin (10 mcg) were placed into agar plates used as positive control and respective solvent was also used as negative control (Fig. 2). After 24 h of incubation, the diameter was observed for zone of inhibition (measured in mm including disc size). Tests were performed in triplicates and observed values of ZOI were expressed as mean value with Standard Error of Means (SEM).
Determination of Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC): All the fractions showing zone of inhibition≥10 mm were tested for the minimum inhibitory concentration to find out the lowest concentration of an extract that inhibits the visible growth of test microorganisms and the same test was used to determine the minimum bactericidal concentration. MIC was performed at five concentrations of extracts (500, 250, 125, 62.5, 31.25 μg) following serial dilution technique. All the tests which did show no visible growth in the MIC test, were subcultured and incubated at 37±1°C overnight. The highest dilution showing 100% (even 99%) inhibition was recorded as MBC.
RESULT
Percentage extract yield for the different solvents used was 7.5 (methanol), 5.3 (hexane), 3.0 (water) and 1.6 (chloroform). The result of screened plant extracts for antibacterial activity is summarized in Table 1.
Methanol fraction showed a variable activity against all the test strains. Out of eight strains tested six strains were significantly inhibited by the methanol extract of V. wallichii. Highest zone of inhibition (20±1.0 mm) was recorded against B. subtilis followed by S. aureus (19±0.8 mm). The lowest activity of methanol extract was observed against E. chrysanthemi and A. tumefaciens (ZOI -9±0.2 mm) each.
Hexane extract was found active against all the tested strains. The highest zone of inhibition (18±1.2 mm) was observed against B. subtilis followed by E. coli (15±1.0 mm). Significant inhibition was also observed against S. aureus (zoi-12±0.2 mm) and E. chrysanthemi (10±0.2 mm) and P. vulgaris (ZOI 10±1.2 mm), however low level of activity was observed against X. phaseoli, A. tumefaciens and X. campestris (<10 mm ZOI).
The aqueous extract showed its highest activity against S. aureus (zoi-23±1.0 mm) followed by B. subtilis (16±1.2 mm). The fraction was also found significantly active against E. coli (13±0.5 mm) and P. vulgaris (13±0.7 mm). A comparatively low level of activity was recorded against E. chrysanthemi (7±0.2 mm) and X. campestris (6±0.2 mm).
Similarly, chloroform extract was also found significantly active against E. coli, S. aureus and P. vulgaris with zone of inhibition of 12±0.5, 10±0.2 and 10±0.2 mm, respectively (Table 1). A very low activity was recorded against B. subtilis. Whereas, it was inactive against remaining strains, which were tested against X. campestris, E. chrysanthemi, A. tumefaciens. It was interesting to note that all the fractions were found more active than the used standard antibiotics i.e. ampicillin and erythromycin (Fig. 3).
MIC/MBC evaluation: A total of 15 tests (having inhibition zone > 10 mm) were performed to determine the MIC/MBC concentration of different extract of V. wallichii aerial part (Table 2).
Of these only one test did not exhibited any inhibition at the tested concentrations for MIC (500, 250, 125, 62.5 and 31.25 μg mL-1). 31.25 μg mL-1 was recorded as MIC value for methanol extract against B. subtilis. As shown in Table 2, MIC value of 125 μg mL-1 was observed in five tests (hexane extract against B. subtilis and P. vulgaris, methanol extract against S. aureus and P. vulgaris, aqueous extract against B. subtilis) and 250 μg mL-1 in nine tests (hexane extract against S. aureus, methanol extract against E. coli and X. campestris, aqueous extract against S. aureus).
| Table 1: | Zone of inhibition of different extracts of V. wallichi aerial part |
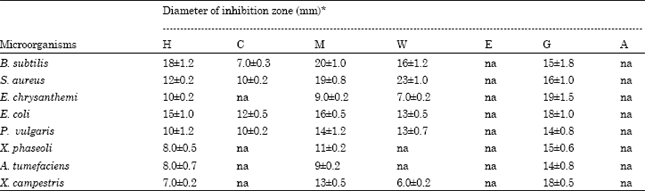 | |
| *All the values are Mean±SEM of three determinations. H, C, M, W: Hexane, Chloroform, Methanol, Aqueous extracts. E, G, A: Erythromycin, Gentamycin, Ampicillin (+control), na: Not active | |
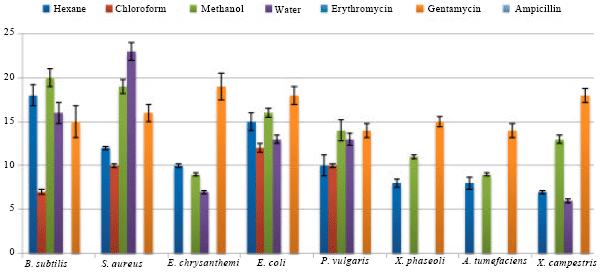 | |
| Fig. 3: | Antibacterial activity of V. wallichi against some bacterial strains |
| Table 2: | Minimum inhibitory/bactericidal/bacteristatic concentration (μg mL-1) |
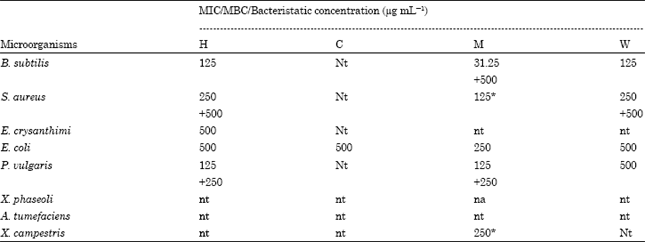 | |
| *: MIC Concentration is also the MBC, +: MBC, nt: Not tested due to lack of significant inhibition (zone of inhibition ≥10 mm) at 1000 μg mL-1. na: No activity observed at tested dilutions unmarked values are MIC values, which are only Bacteristatic | |
In 12 instances (hexane fraction against E. chrysanthemi and E. coli, chloroform extract against E. coli, aqueous extract against E. coli, P. vulgaris) 500 μg mL-1 concentration was found as MIC. As evident from Table 2 that in bactericidal concentration analysis, MIC value of 125 μg mL-1 for methanol extract against S. aureus and 250 μg mL-1 for methanol extract against X. campestris were also exhibited bactericidal effect. However, in 8 tests where the bactericidal activity was not observed for 5 concentrations, therefore their MIC values were recorded as bacteristatic.
DISCUSSION
The high altitude grown Himalayan plant V. wallichii has not been investigated for its defined antimicrobial potentiality. The available literature indicates that V. wallichii is well known for its use in the treatment of various ailments. Though it has been exploited for its various uses by a number of workers all over the world but this indigenous plant has not been studied adequately as antibacterial agents. Therefore, this study highlights for the first time the ability of different solvent fractions of the plant as antibacterial through in vitro assays.
In some recent literatures, ethanol and methanol are used as extractant, however it may not demonstrate the greatest sensitivity in yielding antimicrobial chemicals (Cowan, 1999). For this reason in the present study four extractant, i.e., hexane, chloroform, methanol and water were used to obtain maximum active compound in the extracts. It is interesting to note that a correlation was observed between the extract yield and antibacterial activity of different fractions. This suggests that in serial extraction, maximum biologically active compounds were solublized with methanol followed by hexane, water and chloroform.
The results obtained in this investigation for the antibacterial activity of V. wallichi using disc diffusion method showed that animal pathogenic strains are more sensitive to tested extracts than plant pathogenic strains (Table 1). This low sensitivity of plant pathogenic bacteria might be due to the fact that plant pathogenic bacteria are continuously evolving against phytochemicals and during this process they might have modified their metabolism or developed some resistant in their genetic element.
The test organisms used in this study are associated with various forms of animal and plant diseases. From a clinical point of view, E. coli causes septicemias and can infect the gall bladder, meninges, surgical wounds, skin lesions and the lungs, especially in debilitate and immunodeficient patients (Black, 1996). Similarly, S. aureus is an important nosocomial and community-acquired pathogen and can infect other tissues when barriers have been breached (e.g., skin or mucosal lining). This leads to furuncles (boils) and carbuncles (a collection of furuncles). Moreover in infants S. aureus infection can cause a severe disease staphylococcal scalded skin syndrome (Curran and Al-Salihi, 1980) and therefore the use of V. walichii may be an effective measure to fight against such infections.
The results of present investigation are not only in agreement with the previous findings of Suri and Thind (1978) and Girgune et al. (1980), who suggest that oil of V. wallichii is effective against animal pathogenic bacteria but also provide new lead as there is no previous record on the antimicrobial activity of V. wallichii against plant pathogenic bacterial strains which are responsible for various plant diseases like crown gall, leaf blight, leaf spot and rot disease.
Some workers reported that plant extracts and oils exert a greater inhibitory activity against Gram+ve bacteria (Smith-Palmer et al., 1998) but the results of this study did show no selectivity towards Gram+ve bacteria.
It was interesting to note that in the present study most of the MIC values were found lower than the MBC values which indicate that the extracts might be bactericidal in action. As evident from the Table 2 lower MIC and MBC values and higher zone of inhibition (Fig. 2) for aqueous and methanol extracts connotes higher solubility of phytoconstituents in the less polar fractions.
Various compounds like borneol, alpha-pinene and beta-pinene, caryophyllene, bornyl acetate and d-limonene, which are also reported from V. wallichii October 8, 2010(Sati et al., 2005) were demonstrated to have definite antimicrobial properties (Tabanca et al., 2001; Vardar et al., 2003). This suggests that these compounds may also contribute to the significant antibacterial activity observed during present study. The essential oils containing terpenes are also reported to possess antimicrobial activity (Dorman and Deans, 2000), which are consistent with the present study. However, the synergistic effects of these active chemicals with other constituents of the essential oils should be taken into consideration to establish the antimicrobial activity.
Relying upon the results of the present investigation it could be suggested that these findings may also be exploited for the remedies of a wide range of microbial diseases of plant as well as animals. There is no doubt that the studied plant may be a good source of future drugs that could be used in the treatment of infection caused by these microbes.
ACKNOWLEDGMENTS
The authors wish to thank Dr. Y.P.S. Pangtey, Professor Emeritus for Plant Identification; Department of Plant Pathology, GBPUA and T, Pantnagar and IMTCC, Chandigarh, for providing bacterial cultures and Department of Botany, DSB Campus, Kumaun University, Nainital for providing necessary facilities to carry out present investigation.
REFERENCES
- Bauer, A.W., W.M.M. Kirby, J.C. Sherris and M. Turck, 1966. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol., 45: 493-496.
CrossRefPubMedDirect Link - Cowan, M.M., 1999. Plant products as antimicrobial agents. Clin. Microbiol. Rev., 12: 564-582.
CrossRefPubMedDirect Link - Curran, J.P. and F.L. Al-Salihi, 1980. Neonatal staphylococcal scalded skin syndrome: Massive outbreak due to an unusual phage type. Pediatrics, 66: 285-290.
Direct Link - Dorman, H.J.D. and S.G. Deans, 2000. Antimicrobial agents from plants: Antibacterial activity of plant volatile oils. J. Appl. Microbiol., 88: 308-316.
CrossRefDirect Link - Hamburger, M. and K. Hostettmann, 1991. Bioactivity in plants: The link between phytochemistry and medicine. Phytochemistry, 30: 3864-3874.
CrossRefDirect Link - Khulbe, K. and S.C. Sati, 2009. Antibacterial activity of Boenninghausenia albiflora Reichb. (Rutaceae). Afr. J. Biotechnol., 8: 6346-6348.
Direct Link - Nascimento, G.G.F., J. Locatelli, P.C. Freitas and G.L. Silva, 2000. Antibacterial activity of plant extracts and phytochemicals on antibiotic-resistant bacteria. Braz. J. Microbiol., 31: 247-256.
CrossRefDirect Link - Owolabi, O.J., E.K.I. Omogbai and O. Obasuyi, 2007. Antifungal and antibacterial activities of the ethanolic and aqueous extract of Kigella africana (Bignoniaceae) stem bark. Afr. J. Biotechnol., 6: 1677-1680.
Direct Link - Sati, S.C., S. Chanotiya and C.S. Mathela, 2005. Comparative investigations on the leaf and root oils of Valeriana wallichii DC from North Western Himalaya. J. Essent. Oil Res., 17: 408-409.
Direct Link - Sati, S.C. and S. Joshi, 2010. Antimicrobial potential of leaf extracts of Juniperus commnis L. from Kumaun Himalaya. Afr. J. Microbiol. Res., 4: 1291-1294.
Direct Link - Smith-Palmer, A., J. Stewart and L. Fyfe, 1998. Antimicrobial properties of plant essential oils and essences against five important food-borne pathogens. Lett. Applied Microbiol., 26: 118-122.
CrossRefPubMedDirect Link - Tabanca, N., N. Kirimer, B. Demirci, F. Demirci and K.H.C. Baser, 2001. Composition and antimicrobial activity of the essential oils of Micromeria cristata subsp. phrygia and the enantiomeric distribution of borneol. J. Agric. Food Chem., 49: 4300-4303.
CrossRefDirect Link - Vardar-Unlu, G., F. Candan, A. Sokmen, D. Daferera and M. Polissiou et al., 2003. Antibacterial and antioxidant activity of the essential oil and methanol extracts of Thymus pectinatus Fisch. Et Mey var. Pectinatus (Lamiaceae). J. Agric. Food Chem., 51: 63-67.
CrossRefDirect Link








