Research Article
Lipase Catalyzed-transesterification of Vegetable Oils by Lipolytic Bacteria
Department of Biotechnology, National Institute of Technology, Raipur-492 010, India
L.S.B. Upadhyay
Department of Biotechnology, National Institute of Technology, Raipur-492 010, India
R. Shrivastava
Department of Biotechnology, National Institute of Technology, Raipur-492 010, India









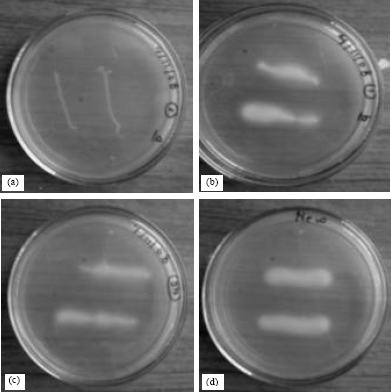
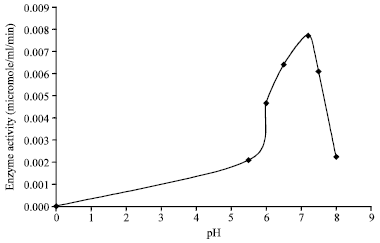
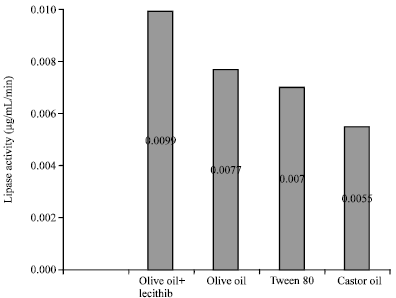
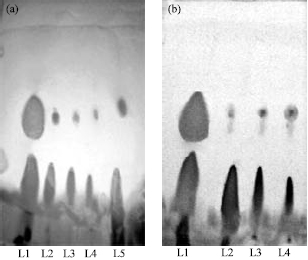
Balaji M. Panchal Reply
Very good research paper in 2011 year