Mini Review
Pyrimidine Base Catabolism in Species of Pseudomonas and Burkholderia
Department of Biology and Microbiology, South Dakota State University, Brookings, SD 57007, USA
The catabolism of the pyrimidine bases uracil and thymine has been shown to provide a source of nitrogen in prokaryotes. It has been established that three different types of pyrimidine catabolic pathways exist in prokaryotes (Vogels and van der Drift, 1976). One pathway involves the oxidative catabolism of uracil and thymine to urea and malonic acid by the enzymes uracil/thymine dehydrogenase, barbiturase and ureidomalonase (Vogels and van der Drift, 1976; Soong et al., 2001, 2002). Strains of Nocardia, Mycobacterium and Enterobacter aerogenes and Rhodococcus erythropolis degrade pyrimidine bases using the oxidative pathway (Hayaishi and Kornberg, 1952; Lara, 1952; Patel and West, 1987; Soong et al., 2001, 2002). A second pathway of pyrimidine catabolism has been recently identified in Escherichia coli K-12 (Loh et al., 2006). It has been found that this pathway degrades uracil and thymine to 3-hydroxypropionic acid and 2-methyl-3-hydroxypropionic acid, respectively and operates at room temperature but not at 37°C (Loh et al., 2006). The third pathway of pyrimidine catabolism is called the reductive pathway and it appears to be more prevalent in prokaryotes than the other types of pyrimidine catabolic pathways (Vogels and van der Drift, 1976).
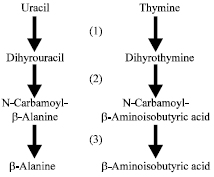 | |
| Fig. 1: | The pyrimidine base reductive catabolic pathway enzymes include (1) dihydropyrimidine dehydrogenase, (2) dihydropyrimidinase and (3) β-ureidopropionase |
The reductive pathway involves three enzymatic steps (Fig. 1). The initial step is catalysed by the enzyme dihydropyrimidine dehydrogenase (EC 1.3.1.2) that converts uracil and thymine to dihydrouracil and dihydrothymine, respectively (Fig. 1). The second pathway step is catalyzed by the enzyme dihydropyrimidinase (EC 3.5.2.2), which converts dihydrouracil and dihydrothymine, respectively, to N-carbamoyl-β-alanine and N-carbamoyl-β-aminoisobutyric acid (Fig. 1). This enzyme usually also has the ability to hydrolyze hydantoins and this could prove vital in the development of large-scale bioreactor systems for the inexpensive production of β-amino acids and D-amino acids (Morin et al., 1986; Chevalier et al., 1989; Sharma and Vohra, 1999; Zhang et al., 2010). The final step of the reductive pathway is catalysed by the enzyme β-ureidopropionase (EC 3.5.1.6). This enzyme produces β-alanine or β-aminoisobutyric acid from their respective N-carbamoyl derivatives (Fig. 1). The reductive pathway operates in a number of prokaryotes including Clostridium uracilicum (Campbell, 1957), Acidovorax facilis (Kramer and Kaltwasser, 1969), Salmonella typhimurium (West et al., 1985) and Escherichia coli B (West, 1998). A number of pseudomonad species, including Pseudomonas aeruginosa (Potter et al., 1982; Kim and West, 1991), Pseudomonas chlororaphis (West, 1991a), Pseudomonas stutzeri (Xu and West, 1992), Pseudomonas fluorescens (Santiago and West, 1999), Pseudomonas putida (West, 2001), Pseudomonas lemonnieri (Burnette et al., 2006) and Pseudomonas syringae (Gant et al., 2007), have been shown to utilize the reductive pathway to degrade pyrimidine bases. The related species Burkholderia cepacia also degraded pyrimidine bases using the reductive pathway (West, 1997). Many of the fluorescent pseudomonads are recognized to be clinically significant in humans. The role of the fluorescent pseudomonads as opportunistic pathogens during the treatment of human cancer with such chemotherapeutic agents as 5-fluorouracil has been noted (Moody et al., 1972). The species P. aeruginosa and B. cepacia are known human pathogens afflicting individuals with cystic fibrosis as well as human burn patients (Eberl and Tummler, 2004). Another pseudomonad of importance is P. syringae, which is a known plant pathogen (Salch and Shaw, 1988). Considering the significance of the pseudomonad species and B. cepacia as pathogens, catabolism of pyrimidine bases was studied in these strains to better understand how these microbes survived under nitrogen-limiting conditions and to identify possible mechanisms of microbiological control.
DIHYDROPYRIMIDINE DEHYDROGENASE
Enzyme function: Dihydropyrimidine dehydrogenase reduces the pyrimidine bases uracil and thymine using a nicotinamide cofactor. Maximal enzyme activity is observed with either NADH or NADPH as a nicotinamide cofactor.
| Table 1: | Nicotinamide cofactor specificity of dihydropyrimidine dehydrogenase from pseudomonads and Burkholderia cepacia |
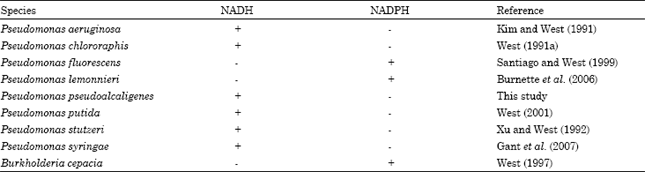 | |
| +: Presence; -: Absence | |
A possible explanation for the pseudomonad dehydrogenase activity being active using either cofactor is likely related to the presence of pyridine nucleotide transhydrogenase (EC 1.6.1.1) in pseudomonad cells (Kaplan, 1955). This enzyme catalyzes a reaction involving the reduction of either NAD+ or NADP+ to synthesize NADH or NADPH (San Pietro et al., 1955; French et al., 1997). NADH is the preferred nicotinamide cofactor for the P. aeruginosa, P. chlororaphis, P. putida, P. stutzeri or P. syringae dihydropyrimidine dehydrogenase (Table 1). NADPH serves as the preferred nicotinamide cofactor for dihydropyrimidine dehydrogenase in P. fluorescens, P. lemonnieri and B. cepacia (Table 1). In P. pseudoalcaligenes ATCC 17740, the preferred nicotinamide cofactor for the dehydrogenase present in cells grown at 30°C with aeration in an orbital shaker (200 revolutions min-1) on 0.2% uracil as a nitrogen source and 0.4% succinate as a carbon source was found to be NADH (T.P. West, unpublished results). The cells were processed and assayed as previously described (West, 1991b). Dehydrogenase specific activity was 2.32 nmol dihydrouracil formed/min/mg protein (within 10% error) using NADH as a cofactor compared to 0.10 nmol dihydrouracil formed/min/mg protein (within 10% error) using NADPH as a cofactor (T.P. West, unpublished results). Similarly, NADH was the preferred nicotinamide cofactor for the dehydrogenase present in cells grown at 30°C with aeration in an orbital shaker (200 revolutions min-1) on 0.2% thymine as a nitrogen source and 0.4% succinate as a carbon source. Dehydrogenase specific activity was 0.97 nmol dihydrouracil formed/min/mg protein (within 10% error) using NADH as a cofactor compared to 0.36 nmol dihydrouracil formed/min/mg protein (within 10% error) using NADPH as a cofactor (T.P. West, unublished results).
Effect of growth conditions: Dihydropyrimidine dehydrogenase activity in species of Pseudomonas and Burkholderia is influenced by the source of nitrogen and carbon. Maximum dehydrogenase activity was observed in P. aeruginosa, P. chlororaphis, P. lemonnieri or P. syringae when the cells were grown on uracil as a nitrogen source and glucose as a carbon source compared to cells grown in a glucose minimal medium containing ammonium sulfate as the nitrogen source (Table 2). In P. pseudoalcaligenes or P. putida, dehydrogenase activity was highest when the cells were grown on uracil as a nitrogen source and succinate as a carbon source relative to cells grown in a succinate minimal medium containing ammonium sulfate as the nitrogen source (Table 2). When P. fluorescens cells were grown on dihydrouracil as a nitrogen source and succinate as a carbon source, maximum dehydrogenase activity was observed compared to cells grown on ammonium sulfate and succinate. Growth on thymine as a nitrogen source and succinate as a carbon source produced the largest increase in dehydrogenase activity in P. stutzeri (Table 2). In B. cepacia, glucose-grown cells produced the highest dehydrogenase activity using 5-methylcytosine as a nitrogen source (Table 2).
| Table 2: | Growth conditions found to produce maximum dihydropyrimidine dehydrogenase activity in species of Pseudomonas species and Burkholderia cepacia |
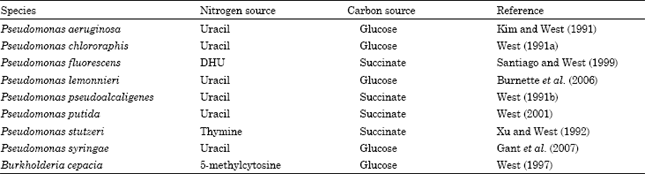 | |
Clearly, the source of nitrogen greatly affected the level of dehydrogenase activity in species of Pseudomonas and Burkholderia.
DIHYDROPYRIMIDINASE
Enzyme function: Relative to the species of Pseudomonas and Burkholderia, only the P. stutzeri dihydropyrimidinase has been characterized from cells growth on dihydrothymine as a nitrogen source and succinate as a carbon source (Xu and West, 1994). The partially purified enzyme has a molecular weight of 115,000 daltons (Xu and West, 1994). The P. stutzeri enzyme was found to utilize dihydrouracil, dihydrothymine and hydantoin as substrates (Xu and West, 1994). The optimal temperature for enzyme activity was 45°C and it was active between pH 7.5-9.0 (Xu and West, 1994). The P. stutzeri enzyme was stimulated by magnesium ions and inhibited by zinc or copper ions (Xu and West, 1994). Although, the dihydropyrimidinase from P. stutzeri could utilize hydantoin as a substrate, it was more specific for the dihydropyrimidine bases as substrates (Xu and West, 1994). Therefore, it appeared that a dihydropyrimidinase activity was present in P. stutzeri although the presence of a D-hydantoinase activity was not investigated.
Effect of growth conditions: Growth on either pyrimidine or dihydropyrimidine bases increased the levels of dihydropyrimidinase activity in the species of Pseudomonas and Burkholderia. Growth of B. cepacia cells on the nitrogen source 5-methylcytosine and the carbon source glucose resulted in the highest observed dihydropyrimidinase activity (Table 3). In P. aeruginosa, growth on uracil as a nitrogen source and glucose as a carbon source produced the highest dihydropyrimidinase activity (Table 3). Succinate-grown cells of P. fluorescens or P. putida exhibited the maximum dihydropyrimidinase activity when thymine served as the nitrogen source (Table 3). In P. pseudoalcaligenes, P. stutzeri or P. syringae, dihydropyrimidinase activity was increased to the highest observed level of activity following cell growth on dihydrothymine as a nitrogen source and succinate as the carbon source (Table 3). Glucose-grown cells of P. lemonnieri produced the highest dihydropyrimidinase activity when dihydrouracil served as the nitrogen source (Table 3). With respect to dihydropyrimidinase activity in P. chlororaphis ATCC 17414, growth of the cells on the nitrogen source dihydrothymine and carbon source glucose resulted in over a 100-fold increase in its activity (Table 3). Using two types of colorimetric assays, it has also been determined that a hydantoin-hydrolyzing activity exists in P. chlororaphis ATCC 17414 in addition to dihydropyrimidinase. When P. chlororaphis ATCC 17414 cells were grown with aeration in an orbital shaker (200 revolutions min-1) at 30°C in a medium containing 0.2% hydantoin as a nitrogen source and 0.4% glucose as a carbon source and the cells were processed and assayed as previously described (West, 1991a), it was possible to detect hydantoinase activity in this strain (T.P. West, unpublished results).
| Table 3: | Growth conditions shown to produce maximum dihydropyrimidinase activity in Pseudomonas species and Burkholderia cepacia |
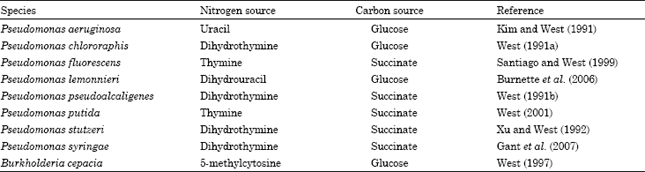 | |
A comparison of dihydropyrimidinase and hydantoinase activities was made in the hydantoin-grown cells at 45°C where 5 mM substrate (dihydrouracil or hydantoin) was included in the reaction mix. Under these conditions, the specific activity of dihydropyrimidinase was <0.06±0.01 nmol/min/mg protein (standard deviation) while the hydantoinase specific activity was 16.68±2.98 nmol/min/mg protein (±standard deviation). Both of these enzyme activities were also assayed in the extracts derived from hydantoin-grown cells at a lower substrate concentration (1 mM) and at a lower assay temperature (30°C). It was determined that the dihydropyrimidinase specific activity was 6.63±0.69 nmol/min/mg protein (±standard deviation) while the hydantoinase specific activity was 0.53±0.07 nmol/min/mg protein (T.P. West, unpublished results). The different activities observed would seem to indicate that the enzymes dihydropyrimidinase and hydantoinase exist independent of one another in P. chlororaphis ATCC 17414 cells. Dihydropyrimidinase activity has also been detected in P. putida (Takahashi et al., 1978; Chevalier et al., 1989; West, 2001). In P. putida strain RU-KM3S, the inactivation of the gene encoding dihydropyrimidinase resulted in the loss of hydantoinase activity suggesting that dihydropyrimidinase was responsible for the observed hydantoinase activity (Matcher et al., 2004). It does not appear that dihydropyrimidinase is solely responsible for hydantoin hydrolysis in Pseudomonas species because a DNA probe from P. putida to detect D-hydantoin-producing microorganisms found that they contained a gene for a D-hydantoinase (LaPointe et al., 1995). The presence of both a dihydropyrimidinase activity and a hydantoinase activity has been confirmed in P. fluorescens (Morin et al., 1986) similar to what was observed in P. chlororaphis cells. It has been found that resting cells of B. cepacia produced a D-hydantoinase from a hydantoin when corn steep liquor served as the nitrogen source (Jiang et al., 2007). Overall, previous work indicates that growth of pseudomonads or B. cepacia on pyrimidine or dihydropyrimidine bases as nitrogen sources resulted in an elevation of dihydropyrimidinase activity that likely exists independently of the hydantoin-hydrolyzing activity.
β-UREIDOPROPIONASE
Enzyme function: A prior study has purified β-ureidopropionase to homogeneity from P. putida IFO12996 (Ogawa and Shimizu, 1994). It was found to have a molecular weight of 90,000 daltons and require the presence of a divalent metal ion for activity (Ogawa and Shimizu, 1994). The enzyme has a broad substrate specificity for N-carbamoyl-α-amino acids and the hydrolysis of the N-carbamoyl-α-amino acids was noted to be L-enantiomer specific (Ogawa and Shimizu, 1994).
| Table 4: | Growth conditions shown to produce maximum β-ureidopropionase in Pseudomonas species and Burkholderia cepacia |
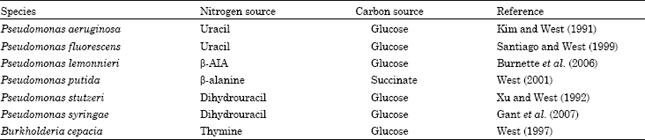 | |
| β-AIA, β-aminoisobutyric acid. | |
The properties of the P. putida β-ureidopropionase were shown to differ from those previously observed in an anaerobic bacterium (Campbell, 1960).
Effect of growth conditio ns: Growth of the species of Pseudomonas and Burkholderia on pyrimidine bases, dihydrouracil, β-alanine and β-aminoisobutyric acid as nitrogen sources increased their cellular levels of β-ureidopropionase activity compared to growth on ammonium sulfate as a nitrogen source. When glucose-grown cells of P. aeruginosa or P. fluorescens utilized uracil as a nitrogen source, β-ureidopropionase was elevated to its highest activity level (Table 4). Similarly, glucose-grown cells of B. cepacia exhibited their highest enzyme activity when thymine served as the nitrogen source (Table 4). The dihydropyrimidine base uracil produced the greatest elevation of β-ureidopropionase activity in glucose-grown cells of P. stutzeri or P. syringae when it served as a nitrogen source (Table 4). In P. lemonnieri, the product of the β-ureidopropionase reaction, namely β-alanine, increased its enzyme activity when succinate-grown cells utilized β-alanine as a source of nitrogen (Table 4). The other product of the β-ureidopropionase reaction, namely β-aminoisobutyric acid, produced the highest enzyme activity in glucose-grown P. putida cells that used β-aminoisobutyric acid as the source of nitrogen (Table 4). Although, dihydropyrimidine dehydrogenase and dihydropyrimidinase activities in the pseudomonads and B. cepacia responded to growth on pyrimidines and dihydropyrimidines as nitrogen sources, it appeared that only β-ureidopropionase activity in pseudomonads was affected by growth on β-alanine or β-aminoisobutyric acid as a nitrogen source (Table 4).
REGULATION AT THE LEVEL OF ENZYME SYNTHESIS
With the pyrimidine catabolic enzyme activities of the species of Pseudomonas and Burkholderia being affected by growth on pyrimidine bases and their catabolic products as nitrogen source, it appeared likely that the catabolic pathway was regulated at the level of enzyme synthesis. In pseudomonads, it has been found that many catabolic pathways of aromatic compounds are subject to regulation by induction of enzyme synthesis by substrates (Ornston and Parke, 1977). Uracil has been found to induce the synthesis of the reductive pathway enzymes in P. aeruginosa and P. fluorescens when glucose served as the carbon source while it was also noted to induce enzyme synthesis in P. putida when succinate served as the carbon source (Table 5). In P. stutzeri, thymine was the inducer of reductive pathway enzyme synthesis when succinate served as the carbon source (Table 5). The inducer of reductive pathway enzyme synthesis in B. cepacia was 5-methylcytosine when glucose was the carbon source (Table 5). Pyrimidine bases were shown to control pyrimidine reductive pathway enzyme synthesis in all prior studies examining species of Pseudomonas and Burkholderia (Kim and West, 1991; Xu and West, 1992; West, 1997; Santiago and West, 1999; West, 2001).
| Table 5: | Growth conditions found to induce pyrimidine catabolic enzyme activities in pseudomonads and Burkholderia cepacia |
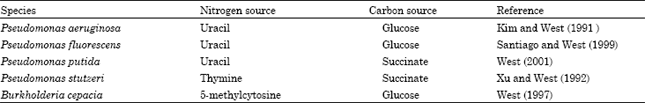 | |
From the prior investigations, it is clear that pyrimidine base catabolism is subject to control at the level of enzyme transcription.
It can be concluded that species of Pseudomonas and Burkholderia actively degrade uracil and thymine using the reductive pathway. It is also clear that the levels of the reductive pathway enzyme activities in these species depend upon the source of nitrogen provided in the growth medium. Further, the three enzyme activities in species of Pseudomonas and Burkholderia are inducible when pyrimidine bases serve as the nitrogen source. It remains to be investigated in these species whether the pyrimidine reductive catabolic pathway consists of only three enzymes or whether it is a pathway consisting of five enzymes. In B. cepacia, 5-methylcytosine has been found to induce the pyrimidine catabolic pathway enzymes. It has been previously shown that the enzyme cytosine deaminase purified from Pseudomonas aureofaciens was capable of deaminating cytosine or 5-methylcytosine to uracil or thymine, respectively (Sakai et al., 1975) and the enzyme is an important pyrimidine salvage enzyme in pseudomonads (Sakai et al., 1976; Beck and O’Donovan, 2008). Cytosine deaminase, which provides the substrates to the dihydropyrimidine dehydrogenase reaction, was induced by 5-methylcytosine in B. cepacia (West, 2000). Similarly, the enzyme β-alanine-pyruvate transaminase that degrades the products of the β-ureidopropionase reaction is induced by 5-methylcytosine in B. cepacia (West, 2000). The ability of 5-methylcytosine to induce the enzymes cytosine deaminase, dihydropyrimidine dehydrogenase, dihydropyrimidinase, β-ureidopropionase and β-alanine-pyruvate transaminase in pseudomonads needs to be more fully explored. It may be that the five enzymes work in unison to degrade the pyrimidine bases that result from nucleic acid catabolism to provide a source of nitrogen for the cells. It also need to be clarified whether species of Pseudomonas and Burkholderia synthesize both dihydropyrimidinase and hydantoinase activities. It may be that the dihydropyrimidinase and hydantoinase are stereospecific to produce D- or L-enantiomers (Syldatk et al., 1999). Each enzyme activity may only be synthesized under specific growth conditions for species of Pseudomonas and Burkholderia. Further, investigations need to determine more fully the role of each enzyme in bacterial metabolism.
This research was supported by the South Dakota Agricultural Experiment Station. The technical assistance of Beth Nemmers during the pyrimidine catabolism studies was greatly appreciated.