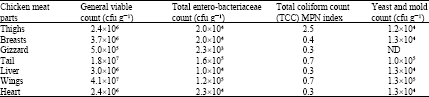
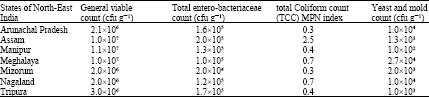
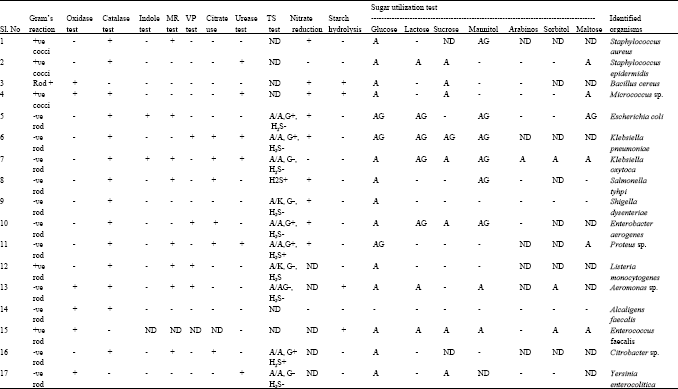
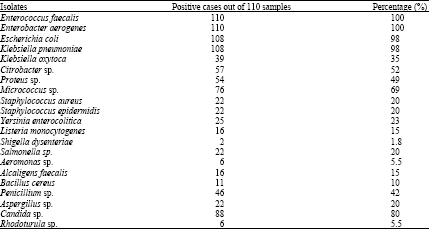
Research Article
Retail Market Poultry Meats of North-East India-A Microbiological Survey for Pathogenic Contaminants
Department of Biotechnology and Bioinformatics, Microbiology Laboratory, North-Eastern Hill University, Shillong, Meghalaya, India
S.R. Joshi
Department of Biotechnology and Bioinformatics, Microbiology Laboratory, North-Eastern Hill University, Shillong, Meghalaya, India