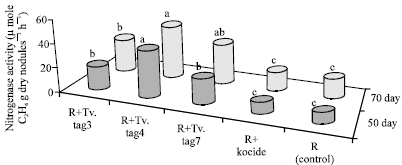
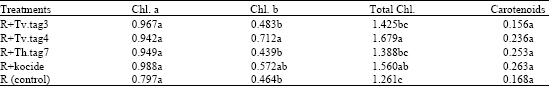
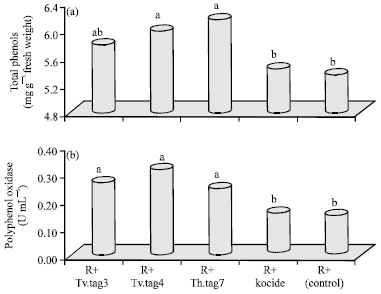
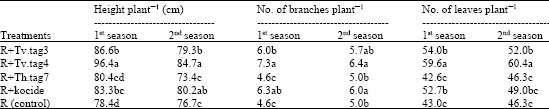
Research Article
Synergistic effect of Trichoderma and Rhizobium on Both Biocontrol of Chocolate Spot Disease and Induction of Nodulation, Physiological Activities and Productivity of Vicia faba
Department of Microbiology, Soils, Water and Environment Research Institute, Agricultural Research Center, Giza, Egypt
K.M. Abd El-Hai
Department of Leguminous and Forage Disease Research,Agricultural Research Center, Giza, Egypt
K.M. Ghoneem
Department of Seed Pathology Research, Plant Pathology Research Institute, Agricultural Research Center, Giza, Egypt